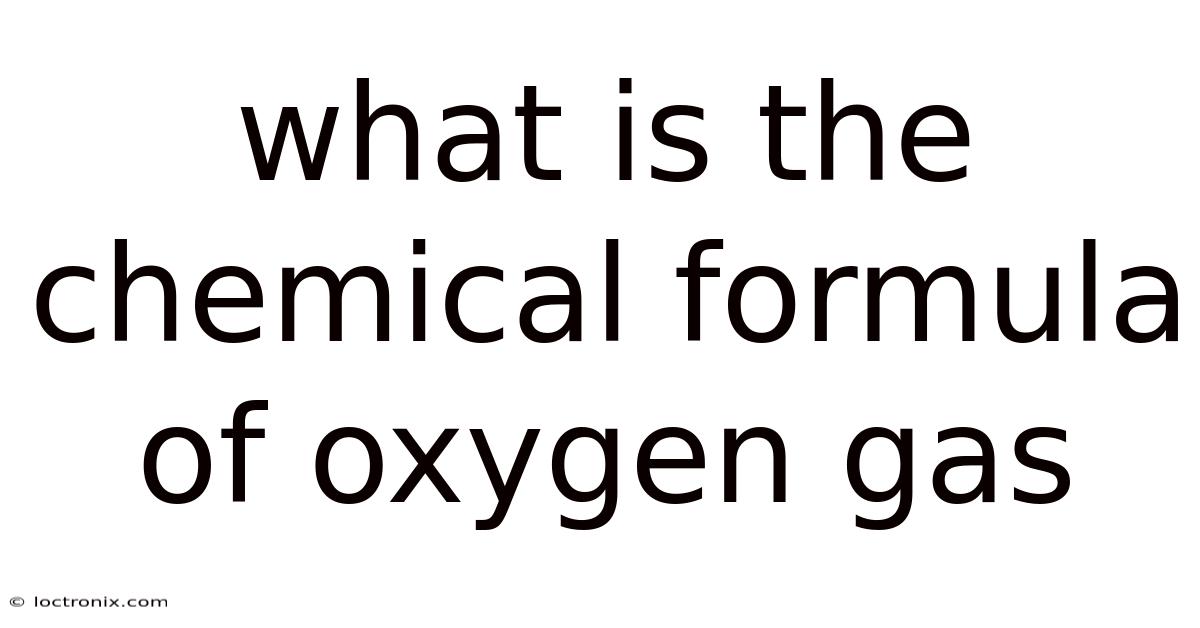##What is the chemical formula of oxygen gas?
The chemical formula of oxygen gas is O₂, representing a diatomic molecule composed of two oxygen atoms bonded together. This simple notation is the cornerstone of countless chemical equations, biological processes, and industrial applications, making it a fundamental concept for students, educators, and anyone curious about the substances that sustain life on Earth Still holds up..
Understanding the Basics
Definition of Oxygen Gas
Oxygen gas refers to the elemental form of oxygen that exists as a colorless, odorless, and tasteless diatomic molecule under standard temperature and pressure (STP). In this state, each molecule contains exactly two oxygen atoms sharing a double covalent bond, which stabilizes the molecule overall.
Why “O₂” and Not “O”?
While the atomic number of oxygen is 8, the element does not naturally exist as isolated atoms under everyday conditions. Instead, two atoms pair up to form O₂, reducing the system’s overall energy and achieving a more stable configuration. This pairing is why the correct chemical formula of oxygen gas is O₂, not simply “O”.
The Molecular Structure of O₂
Bonding and Electron Configuration
Each oxygen atom has six valence electrons. In an O₂ molecule, two of these electrons from each atom overlap to form a double bond, consisting of one sigma (σ) bond and one pi (π) bond. This bonding arrangement satisfies the octet rule for both atoms, resulting in a low‑energy, stable molecule Worth keeping that in mind. Surprisingly effective..
Molecular Geometry
The geometry of an O₂ molecule is linear, with the two oxygen atoms aligned along a straight axis. The bond length is approximately 1.21 Å (angstroms), and the bond angle is 180°, reflecting the simple yet elegant structure of the diatomic molecule It's one of those things that adds up. Nothing fancy..
How Oxygen Gas Appears in the Natural World
Atmospheric Presence
Approximately 21% of Earth’s atmosphere consists of oxygen gas, making it the second most abundant component after nitrogen. This proportion is relatively constant, thanks to the balanced cycles of photosynthesis and respiration that continuously replenish and consume O₂.
Role in Biological Systems
In living organisms, O₂ serves as the final electron acceptor in cellular respiration, enabling the production of adenosine triphosphate (ATP), the energy currency of cells. Without the chemical formula of oxygen gas—O₂—the biochemical pathways that power muscles, brains, and every cellular function would be impossible That's the part that actually makes a difference. Practical, not theoretical..
Occurrence in Water and Minerals
Although water (H₂O) contains oxygen, it is chemically bound to hydrogen. In minerals such as silicates, oxygen atoms occupy lattice positions, but the elemental form remains O₂ when it exists as a discrete gas Worth keeping that in mind..
Chemical Properties and Common Reactions
Combustion
One of the most familiar uses of O₂ is in combustion reactions, where it reacts with fuels to produce heat and light. Here's one way to look at it: the burning of methane (CH₄) can be represented as:
CH₄ + 2 O₂ → CO₂ + 2 H₂O + heat.
Here, the chemical formula of oxygen gas is essential for balancing the equation and understanding energy release.
Oxidation
Oxidation processes involve the transfer of electrons to O₂, often resulting in the formation of oxides. Rust (Fe₂O₃) forms when iron reacts with O₂ and water:
4 Fe + 3 O₂ → 2 Fe₂O₃. Such reactions illustrate how the chemical formula of oxygen gas participates in both slow and rapid transformations.
Acid‑Base Interactions
When O₂ dissolves in water, it can form carbonic acid through reaction with dissolved carbon dioxide, influencing ocean pH levels. While not a direct reaction with O₂, understanding the solubility and reactivity of O₂ aids in comprehending these environmental impacts Took long enough..
Common Misconceptions
“Oxygen is a single atom”
A frequent error is to refer to oxygen simply as “O”. In reality, the elemental form that we breathe is O₂, a molecule of two atoms. Isolated oxygen atoms (O) are highly reactive radicals that quickly recombine to form O₂ Not complicated — just consistent..
“All forms of oxygen are the same”
Oxygen exists in several allotropic forms, including ozone (O₃) and singlet oxygen (¹O₂). While O₂ is the most stable and prevalent, these other forms have distinct properties and applications, such as in disinfection (ozone) and photochemical research (singlet oxygen).
Frequently Asked Questions
What is the chemical formula of oxygen gas?
The chemical formula of oxygen gas is O₂, indicating a molecule composed of two oxygen atoms bonded together Most people skip this — try not to..
Why is O₂ paramagnetic?
O₂ contains two unpaired electrons in its molecular orbital configuration, giving it weak magnetic properties. This paramagnetism can be demonstrated experimentally with a simple magnet Simple as that..
How is oxygen gas produced industrially?
Industrial production typically involves fractional distillation of liquefied air, where O₂ is separated from nitrogen and other gases based on differing boiling points.
Can oxygen exist as a single atom? Yes, but only under extreme conditions such as high-energy plasma or in outer space. In everyday environments, O₂ remains the dominant form.
Is O₂ the same as “oxygen” in everyday language?
In common usage, “oxygen” often refers to the gas we inhale, which is indeed O₂. On the flip side, in chemical contexts, precise terminology distinguishes O₂ from other oxygen‑containing species.
Conclusion
The chemical formula of oxygen gas—O₂—encapsulates a simple yet profound concept that underpins life, industry, and the very atmosphere we inhabit. Because of that, from the double covalent bond that stabilizes the molecule to its important role in respiration, combustion, and oxidation, O₂ is a versatile and indispensable player in the chemical world. By grasping its molecular structure, natural abundance, and reactive behavior, learners can appreciate how a single notation conveys a wealth of scientific meaning. Whether you are a student preparing for an exam, a teacher crafting a lesson, or a curious reader, recognizing the significance of O₂ opens the door to deeper exploration of chemistry and the natural phenomena that shape our universe.
The official docs gloss over this. That's a mistake Small thing, real impact..
Environmental and Technological Implications
The prevalence of O₂ in the atmosphere has far‑reaching consequences beyond biology. Here's the thing — in the context of climate change, the balance between O₂ and oxidized carbon species (CO₂, carbonates) dictates the long‑term sequestration of carbon. When organic matter is buried and subjected to high pressure and temperature, O₂ is effectively removed from the system, allowing carbon to be locked away as fossil fuels. Conversely, the combustion of those fuels re‑introduces O₂‑derived CO₂ into the atmosphere, completing a cycle that drives greenhouse‑gas concentrations Which is the point..
In the realm of energy storage, O₂ is a key reactant in metal‑air batteries. g.Lithium‑air and zinc‑air cells rely on the reversible reduction of O₂ at the cathode to form peroxide or oxide species, delivering energy densities that rival or exceed conventional lithium‑ion technology. Understanding the kinetics of O₂ reduction, the formation of solid‑electrolyte interphases, and the management of side reactions (e., formation of singlet oxygen) is essential for scaling these batteries from laboratory prototypes to commercial products It's one of those things that adds up..
Easier said than done, but still worth knowing.
Safety Considerations
While O₂ is essential for life, its high concentration can be hazardous. Environments enriched with O₂ (>23 % by volume) dramatically accelerate combustion; materials that are normally flame‑retardant can ignite spontaneously. Worth adding: this principle underlies stringent safety protocols in aerospace, medical, and industrial settings where pure or enriched oxygen is stored or used. Proper ventilation, compatible materials, and spark‑free equipment are mandatory to prevent fires and explosions.
Emerging Research Frontiers
-
Catalytic Activation of O₂ – Transition‑metal complexes that can selectively activate O₂ under mild conditions are being explored for green oxidation reactions. By mimicking enzymatic pathways (e.g., cytochrome P450), chemists aim to replace stoichiometric oxidants with O₂, reducing waste and improving sustainability.
-
Artificial Photosynthesis – Replicating the natural splitting of water into O₂ and H₂ is a cornerstone of renewable‑energy research. Photocatalysts that efficiently evolve O₂ while harvesting solar energy could provide a clean source of both oxygen and hydrogen fuel That's the part that actually makes a difference..
-
Space Exploration – In situ resource utilization (ISRU) on Mars and the Moon envisions extracting O₂ from regolith or atmospheric CO₂. Technologies such as solid‑oxide electrolysis and plasma‑based oxygen extraction are being tested to support life‑support systems and rocket propellant production beyond Earth.
Integrating O₂ Into the Broader Chemical Narrative
When teaching or learning chemistry, it is useful to view O₂ not as an isolated fact but as a node in a network of concepts:
- Periodic Trends – Oxygen’s position in group 16 explains its high electronegativity and tendency to form both covalent and ionic bonds.
- Molecular Orbital Theory – The paramagnetism of O₂ provides a concrete example of how electron configuration predicts magnetic behavior.
- Thermodynamics – The exothermic nature of oxidation reactions illustrates the flow of energy from chemical bonds to heat and work.
- Kinetics – The activation energy required for O₂ to react with various substrates underscores the role of catalysts in everyday processes.
By linking the simple formula O₂ to these larger ideas, educators can help students develop a cohesive mental model of chemistry, where each symbol is a gateway to a suite of interrelated phenomena.
Final Thoughts
The succinct notation O₂ belies the molecule’s extraordinary versatility. Also, from sustaining aerobic life and powering engines to enabling cutting‑edge energy storage and driving planetary cycles, diatomic oxygen is a cornerstone of both the natural world and modern technology. On the flip side, recognizing its molecular structure, reactivity, and the contexts in which it operates equips us to harness its benefits responsibly while mitigating its risks. As research continues to get to new ways to manipulate and employ O₂—whether through greener catalytic processes, advanced battery chemistries, or extraterrestrial resource extraction—the humble diatomic molecule will remain at the heart of scientific innovation and environmental stewardship.
Most guides skip this. Don't.