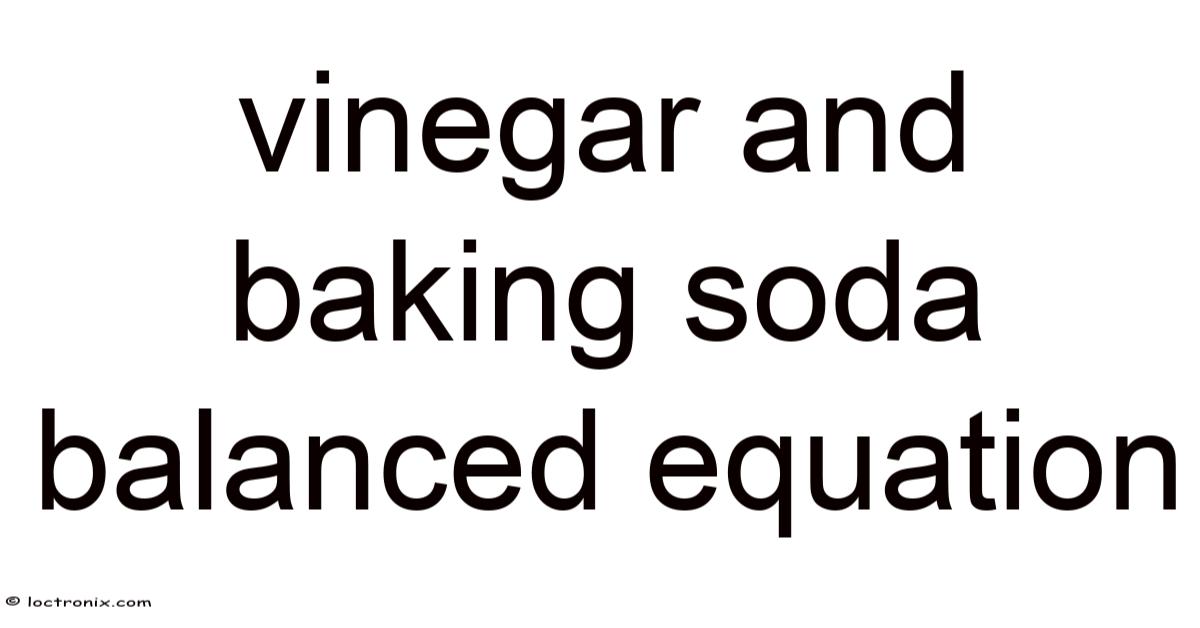The Vinegarand Baking Soda Balanced Equation: A Complete Guide
The vinegar and baking soda balanced equation represents a classic acid‑base reaction that produces carbon dioxide gas, water, and sodium acetate, illustrating fundamental principles of chemistry in a simple household experiment. This reaction is widely used in educational settings, science fairs, and everyday demonstrations because it combines readily available substances to generate observable effects such as fizzing and bubbling. Understanding the equation, the steps involved, and the underlying scientific concepts can deepen learners’ appreciation for chemical changes and the law of conservation of mass.
Introduction
When vinegar (an aqueous solution of acetic acid, CH₃COOH) is mixed with baking soda (solid sodium bicarbonate, NaHCO₃), an immediate effervescent reaction occurs. The balanced chemical equation for this process is:
CH₃COOH + NaHCO₃ → CH₃COONa + H₂O + CO₂↑
This equation shows that one molecule of acetic acid reacts with one molecule of sodium bicarbonate to form one molecule of sodium acetate, one molecule of water, and one molecule of carbon dioxide gas, which escapes as bubbles. The reaction exemplifies a neutralization process where an acid (vinegar) reacts with a base (baking soda) to produce a salt, water, and a gaseous product Easy to understand, harder to ignore..
Not obvious, but once you see it — you'll see it everywhere.
Steps to Perform the Reaction
-
Gather Materials
- White distilled vinegar (5 % acetic acid)
- Baking soda (sodium bicarbonate)
- A clear container or flask (e.g., a glass beaker)
- Measuring cups or a graduated cylinder
- Optional: food coloring or dish soap for visual effects
-
Measure the Reactants
- Pour 50 mL of vinegar into the container.
- In a separate small cup, measure 1 teaspoon (≈5 g) of baking soda.
-
Initiate the Reaction
- Quickly add the baking soda to the vinegar while stirring gently.
- Observe the rapid formation of bubbles as carbon dioxide gas is released.
-
Capture the Products (Optional)
- If you wish to collect the sodium acetate solution, allow the reaction to complete, then filter the mixture to separate any undissolved baking soda.
- The filtrate contains sodium acetate, a useful ingredient in cooking and food preservation.
-
Safety Considerations
- Perform the experiment in a well‑ventilated area to avoid inhaling large amounts of carbon dioxide.
- Avoid contact with eyes; rinse immediately with water if splashes occur.
Scientific Explanation
Acid‑Base Neutralization
The vinegar and baking soda reaction is a neutralization process, a type of double displacement reaction where an acid (CH₃COOH) reacts with a base (NaHCO₃). In this context, the acid donates a proton (H⁺) to the bicarbonate ion (HCO₃⁻), forming carbonic acid (H₂CO₃), which is unstable and rapidly decomposes into water (H₂O) and carbon dioxide (CO₂) Not complicated — just consistent..
Stepwise Mechanism
-
Proton Transfer:
[ \text{CH}_3\text{COOH} + \text{NaHCO}_3 \rightarrow \text{CH}_3\text{COO}^- + \text{Na}^+ + \text{H}_2\text{CO}_3 ] -
Decomposition of Carbonic Acid:
[ \text{H}_2\text{CO}_3 \rightarrow \text{H}_2\text{O} + \text{CO}_2\uparrow ] -
Formation of Sodium Acetate:
The remaining ions, sodium (Na⁺) and acetate (CH₃COO⁻), combine to form sodium acetate (CH₃COONa), which stays dissolved in the solution.
Conservation of Mass
The balanced equation guarantees that the number of atoms of each element is conserved on both sides of the reaction:
- Carbon (C): 2 atoms on the left (one in CH₃COOH, one in NaHCO₃) → 2 atoms on the right (one in CH₃COONa, one in CO₂).
- Hydrogen (H): 5 atoms on the left (4 in CH₃COOH, 1 in NaHCO₃) → 5 atoms on the right (3 in CH₃COONa, 2 in H₂O).
- Oxygen (O): 5 atoms on the left (2 in CH₃COOH, 3 in NaHCO₃) → 5 atoms on the right (2 in CH₃COONa, 1 in H₂O, 2 in CO₂).
This adherence to the law of conservation of mass confirms the correctness of the balanced equation Most people skip this — try not to. And it works..
Observable Effects
The rapid evolution of CO₂ gas creates the characteristic fizzing sound and visual bubbles. If dish soap is added, the soap reduces surface tension, trapping the gas in foam and producing a voluminous “volcano” effect. The temperature change is modest; the reaction is endothermic, absorbing a small amount of heat from the surroundings, which may make the mixture feel slightly cooler.
FAQ
Q1: Why is the equation written with a upward arrow (↑) next to CO₂?
A: The upward arrow indicates that carbon dioxide is a gas that escapes from the reaction mixture, emphasizing its physical state change.
Q2: Can the reaction be reversed to regenerate vinegar and baking soda?
A: No. The products (sodium acetate, water, and carbon dioxide) are chemically stable under normal conditions; reversing the reaction would require additional energy and different reactants, which is not practical in a household setting.
Q3: How does the concentration of vinegar affect the reaction rate?
A: Higher concentrations of acetic acid increase the number of acid molecules available to react, thereby accelerating the rate of proton transfer and gas production. That said, the balanced equation remains the same; only the reaction speed changes.
**Q4: Is sodium acetate safe to
Q4: Is sodium acetate safe to handle? A: The salt is non‑toxic at the concentrations produced in the experiment, but it is mildly basic and may irritate the skin or eyes if it comes into direct contact. Rinsing with water after accidental exposure eliminates any discomfort. In larger quantities it can be used as a buffer in laboratory preparations, confirming its benign profile when managed responsibly.
Practical Applications
Beyond the visual spectacle, the reaction illustrates principles that extend into several fields:
- Food industry – Sodium acetate serves as a preservative and flavor enhancer, while the controlled release of CO₂ is exploited in carbonated beverages and leavening agents.
- Pharmaceuticals – Buffered solutions containing acetate maintain a stable pH, which is crucial for the efficacy of certain drug formulations.
- Environmental science – The same acid‑base chemistry is employed to neutralize acidic runoff in water treatment, demonstrating how a simple household reaction can be scaled for larger‑scale remediation.
Energy Considerations
Although the process absorbs a small amount of heat, the overall enthalpy change is modest. The endothermic nature becomes evident when the mixture feels noticeably cooler to the touch, especially when performed in a sealed container where heat cannot escape readily. This subtle temperature shift underscores the fact that chemical transformations are not always accompanied by dramatic temperature spikes; sometimes the energy exchange is so gentle that it is only perceptible with sensitive instrumentation.
Worth pausing on this one It's one of those things that adds up..
Extending the Experiment
Enthusiasts often modify the classic demonstration to explore related phenomena:
- Variable acid strength – Substituting citric acid or lemon juice for vinegar alters the reaction rate, offering a visual comparison of how different carboxylic acids behave.
- Alternative bases – Using potassium bicarbonate instead of sodium bicarbonate yields potassium acetate, which crystallizes more readily and can be harvested for further study.
- Temperature effects – Conducting the reaction in an ice bath slows the effervescence, while heating the mixture accelerates gas evolution, providing a hands‑on illustration of temperature‑dependent kinetics.
These variations reinforce the underlying stoichiometry while encouraging curiosity about how subtle changes in reactants influence observable outcomes.
Conclusion The decomposition of sodium bicarbonate in the presence of acetic acid exemplifies a harmonious blend of simplicity and scientific depth. A balanced chemical equation not only guarantees that matter is conserved but also serves as a roadmap for predicting products, visualizing gas release, and understanding the subtle energy shifts that accompany the transformation. By dissecting each step — from proton transfer to carbonic‑acid breakdown — students gain insight into fundamental acid‑base concepts, while the vivid fizzing provides an unforgettable sensory cue.
Also worth noting, the reaction’s modest scale belies its broader relevance: the same chemistry underpins food preservation, pharmaceutical buffering, and environmental remediation. Recognizing these connections transforms a classroom demonstration into a gateway for appreciating how everyday materials can be harnessed to solve real‑world challenges That alone is useful..
In sum, the fizzing reaction is more than a party trick; it is a compact laboratory that encapsulates core principles of stoichiometry, thermodynamics, and applied chemistry. When approached with careful observation and thoughtful inquiry, it continues to inspire both novices and seasoned scientists, reminding us that profound knowledge often emerges from the most unassuming of experiments.
You'll probably want to bookmark this section It's one of those things that adds up..