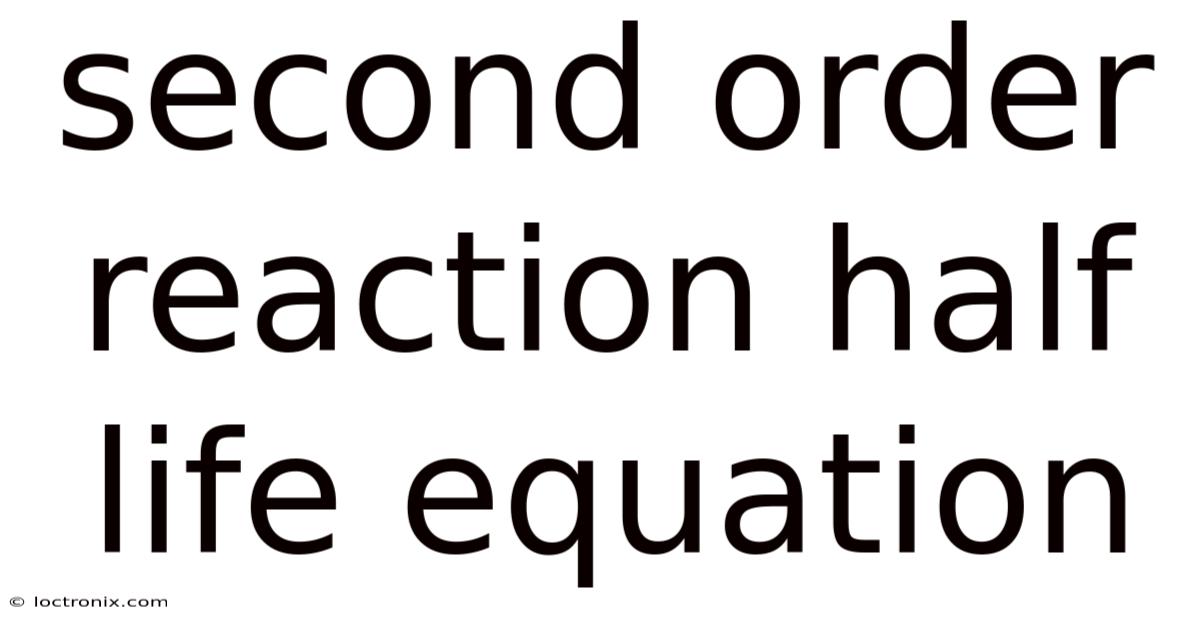Second order reaction half life equation is a cornerstone concept in chemical kinetics that allows scientists and students to predict how long a reaction will take to reach half of its initial concentration. Understanding this equation not only clarifies the behavior of reactants in a variety of natural and industrial processes but also provides a solid foundation for more advanced topics such as reaction mechanisms and rate laws. In this article we will explore the theoretical basis, derive the key formulas, examine real‑world examples, and address frequently asked questions, all while keeping the explanation clear and accessible.
What Defines a Second‑Order Reaction?
A reaction is classified as second order when the overall rate depends on the square of the concentration of a single reactant or on the product of the concentrations of two reactants. Mathematically, for a single reactant A the rate law can be expressed as:
- Rate = k[A]² (when two molecules of A collide)
- Rate = k[A][B] (when A and B react together)
Here, k represents the rate constant, which is specific to each reaction at a given temperature. The order of the reaction is determined experimentally, not from the stoichiometric coefficients of the balanced equation Turns out it matters..
Key Characteristics
- Rate constant units: For a second‑order reaction, k typically has units of L·mol⁻¹·s⁻¹ (or M⁻¹·s⁻¹).
- Concentration dependence: Doubling the concentration of a reactant quadruples the reaction rate.
- Integrated rate law: The concentration of the reactant decreases in a hyperbolic fashion over time, unlike the linear decay seen in first‑order reactions.
Deriving the Half‑Life Equation for a Second‑Order Reaction
The half‑life (t₁/₂) is defined as the time required for the concentration of a reactant to fall to 50 % of its initial value. For a second‑order reaction involving a single reactant A, the integrated rate law is:
[ \frac{1}{[A]} = \frac{1}{[A]_0} + kt ]
where [A]₀ is the initial concentration and [A] is the concentration at time t. To find the half‑life, set [A] = ½[A]₀ and solve for t:
[ \frac{1}{\frac{1}{2}[A]_0} = \frac{1}{[A]0} + kt{1/2} ]
Simplifying the left side gives 2/[A]₀, so:
[ \frac{2}{[A]_0} = \frac{1}{[A]0} + kt{1/2} ]
Subtract 1/[A]₀ from both sides:
[ \frac{1}{[A]0} = kt{1/2} ]
Finally, solve for t₁/₂:
[ \boxed{t_{1/2} = \frac{1}{k[A]_0}} ]
Important takeaway: The half‑life of a second‑order reaction is inversely proportional to both the rate constant k and the initial concentration [A]₀. This contrasts sharply with first‑order reactions, where t₁/₂ is independent of concentration.
Example Calculation
Suppose a reaction has a rate constant k = 0.Which means 025 L·mol⁻¹·s⁻¹ and an initial concentration [A]₀ = 0. 10 M And it works..
[ t_{1/2} = \frac{1}{(0.025)(0.10)} = \frac{1}{0 The details matter here..
If the initial concentration is doubled to 0.20 M, the half‑life becomes 200 s, illustrating the concentration dependence.
Practical Applications
1. Pharmacokinetics
In drug elimination, many elimination pathways follow second‑order kinetics, especially when the drug is metabolized by enzymes that become saturated. Knowing t₁/₂ helps clinicians schedule dosing intervals The details matter here..
2. Environmental Chemistry
The degradation of pollutants such as certain pesticides often follows second‑order kinetics in aqueous environments. Predicting half‑life assists in assessing how long a contaminant will persist.
3. Polymer Chemistry
Chain‑growth polymerizations can exhibit second‑order termination steps. The half‑life of active chain ends influences the molecular weight distribution of the final polymer.
Factors That Influence the Half‑Life| Factor | Effect on t₁/₂ | Explanation |
|--------|------------------|-------------| | Rate constant (k) | ↑k → ↓t₁/₂ | A larger k means a faster reaction, shortening the half‑life. | | Initial concentration ([A]₀) | ↑[A]₀ → ↓t₁/₂ | Because t₁/₂ ∝ 1/[A]₀, higher starting concentrations accelerate the decay. | | Temperature | ↑T → ↑k (usually) | According to the Arrhenius equation, raising temperature increases k, thus reducing t₁/₂. | | Presence of catalysts | ↑catalyst → ↑k | Catalysts lower the activation energy, increasing k and shortening half‑life. |
Common Misconceptions
-
Misconception: Half‑life is constant for all reactions.
Reality: Only first‑order reactions have a constant half‑life. For second‑order reactions, t₁/₂ changes as the concentration changes. -
Misconception: The half‑life equation works for any order.
Reality: The derived formula t₁/₂ = 1/(k[A]₀) is specific to second‑order kinetics. Different orders require distinct expressions Simple as that.. -
Misconception: If you double the initial concentration, the half‑life stays the same. Reality: Doubling *[A]₀ halves the half‑life, as shown by the inverse relationship And that's really what it comes down to..
Frequently Asked Questions (FAQ)
Q1: Can the half‑life equation be used for reactions involving two different reactants?
**
Reactions Involving Two Different Reactants
The half-life formula for second-order kinetics, t₁/₂ = 1/(k [A]₀), is specifically derived for reactions where a single reactant (A) is consumed in a second-order process (e.g., 2A → products or A + B → products where [B] is held constant). That said, the scenario changes significantly when the reaction involves two distinct reactants (A and B) and the initial concentrations of both are different and not held constant Most people skip this — try not to..
The General Formula for Two Reactants:
For a reaction like A + B → products following second-order kinetics overall, the rate law is:
rate = k [A] [B]
The integrated rate law is more complex than for a single reactant. The half-life depends on the initial concentrations of both A and B. The formula is:
t₁/₂ = 1 / (k [A]₀ [B]₀)
Key Implications:
- Both Concentrations Matter: You must know the initial concentration of both reactants ([A]₀ and [B]₀) to calculate the half-life. The formula is the product of the two concentrations.
- Different Initial Concentrations: If [A]₀ ≠ [B]₀, the half-life is determined by the product of these two specific values. Doubling only one concentration does not simply halve the half-life.
- Example Calculation:
- Consider A + B → products with k = 0.025 L·mol⁻¹·s⁻¹.
- If [A]₀ = 0.10 M and [B]₀ = 0.20 M, then: t₁/₂ = 1 / (0.025 * 0.10 * 0.20) = 1 / (0.0005) = 2000 s
- If [A]₀ = 0.20 M and [B]₀ = 0.10 M (swapping the initial concentrations), then: t₁/₂ = 1 / (0.025 * 0.20 * 0.10) = 1 / (0.0005) = 2000 s
- Notice that while the individual concentrations changed, the product [A]₀[B]₀ remained the same (0.020 M²), resulting in the same half-life. Even so, if you only doubled [A]₀ (from 0.10 to 0.20 M) while keeping [B]₀ constant at 0.20 M, the product would be 0.040 M², and the half-life would halve to 1000 s.
- Pseudo-First-Order Behavior: If one reactant is present in a large excess compared to the other (e.g., [B] >> [A]), the reaction can often be treated as pseudo-first-order with respect to A. In this case, the half-life for A becomes approximately constant (t₁/₂ = 1/(k' [A
₀)), where k' is a constant representing the rate constant for the dominant reaction pathway. This simplification is useful in many practical applications, but make sure to remember that it's an approximation valid only under specific conditions.
Conclusion
The half-life equation, t₁/₂ = 1/(k [A]₀), provides a powerful tool for understanding the rate of reactions involving a single reactant. Even so, its application extends beyond simple single-reactant scenarios. Think about it: when dealing with reactions involving two different reactants, the half-life calculation becomes more detailed, requiring the product of the initial concentrations of both reactants. Consider this: understanding the implications of different initial concentrations and the concept of pseudo-first-order behavior are crucial for accurate prediction and control of reaction rates. Now, by considering these factors, chemists and engineers can effectively make use of the half-life equation to optimize chemical processes and achieve desired outcomes. It underscores the importance of carefully analyzing reaction kinetics and considering all relevant variables when applying this fundamental principle of chemical kinetics.