Oxidation Number Of Iron In Fe2o3
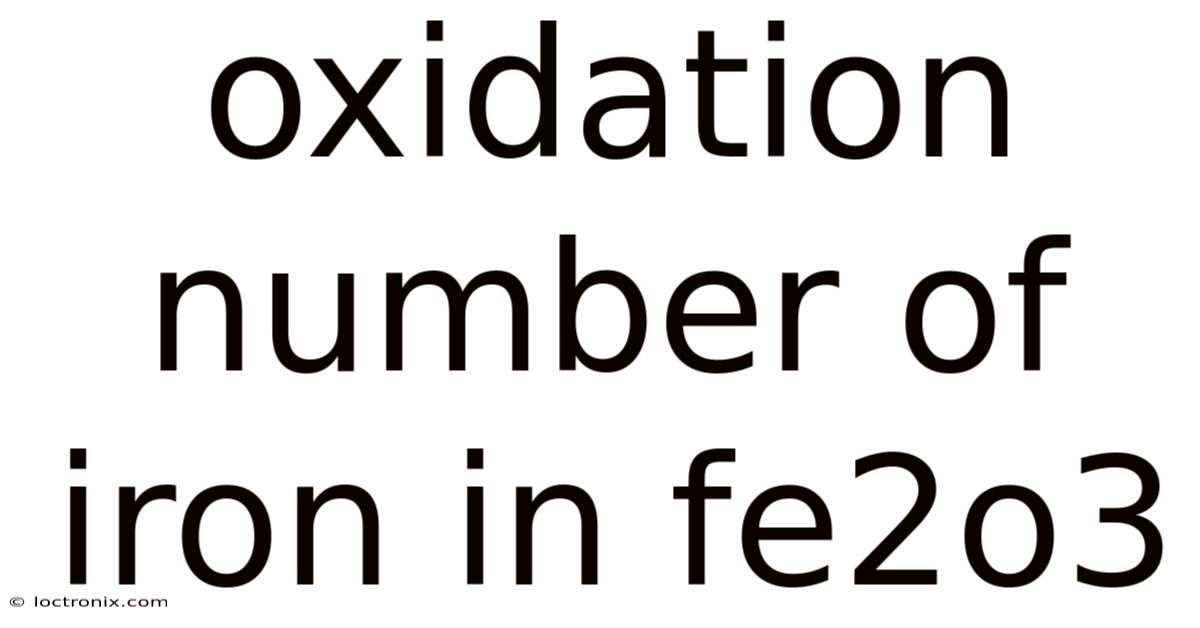
Iron(III) oxide, commonly known as rust or Fe₂O₃, is a compound that plays a significant role in chemistry, metallurgy, and everyday life. Understanding the oxidation number of iron in this compound is crucial for students, researchers, and professionals dealing with redox reactions, corrosion, or material science. This article will explore the oxidation state of iron in Fe₂O₃, explain the underlying principles, and provide practical examples to solidify understanding.
Introduction
Oxidation numbers are a way of representing the degree of oxidation of an atom in a chemical compound. In Fe₂O₃, determining the oxidation number of iron requires understanding the rules for assigning oxidation states and the nature of the compound itself. This article will guide you through the process, explain the significance of the oxidation number, and answer common questions about iron's oxidation states.
What is the Oxidation Number of Iron in Fe₂O₃?
The oxidation number of iron in Fe₂O₃ is +3. This means that each iron atom in the compound has lost three electrons, resulting in a positive charge of three. The compound Fe₂O₃ consists of two iron atoms and three oxygen atoms. Since oxygen typically has an oxidation number of -2, the total negative charge from the three oxygen atoms is -6. To balance this, the two iron atoms together must have a total oxidation number of +6, which means each iron atom has an oxidation number of +3.
How to Determine the Oxidation Number of Iron in Fe₂O₃
To find the oxidation number of iron in Fe₂O₃, follow these steps:
- Identify the known oxidation numbers: Oxygen usually has an oxidation number of -2 in compounds, except in peroxides or when bonded to fluorine.
- Set up the equation: Let the oxidation number of iron be x. Since there are two iron atoms and three oxygen atoms, the equation is: $2x + 3(-2) = 0$
- Solve for x: $2x - 6 = 0$ $2x = 6$ $x = +3$
Therefore, the oxidation number of iron in Fe₂O₃ is +3.
Why is the Oxidation Number of Iron in Fe₂O₃ +3?
The oxidation number of iron in Fe₂O₃ is +3 because of the way electrons are distributed in the compound. Iron can have multiple oxidation states, but in Fe₂O₃, it has lost three electrons to achieve a stable electronic configuration. Oxygen, being highly electronegative, attracts electrons from iron, resulting in iron's oxidation number of +3 and oxygen's -2.
The Significance of Iron's Oxidation Number in Fe₂O₃
Understanding the oxidation number of iron in Fe₂O₃ is important for several reasons:
- Redox Reactions: In redox reactions, the change in oxidation number indicates whether an element is oxidized or reduced.
- Corrosion: Rust formation is a common example of iron being oxidized from Fe to Fe³⁺.
- Material Properties: The oxidation state affects the color, magnetic properties, and reactivity of iron compounds.
Comparison with Other Iron Oxides
Iron can form several oxides with different oxidation numbers:
- FeO (Iron(II) oxide): Iron has an oxidation number of +2.
- Fe₂O₃ (Iron(III) oxide): Iron has an oxidation number of +3.
- Fe₃O₄ (Magnetite): A mixed oxide where iron has both +2 and +3 oxidation states.
This variety highlights the versatility of iron in forming compounds with different properties.
Practical Examples and Applications
- Rust Formation: When iron corrodes in the presence of oxygen and moisture, it forms Fe₂O₃, where iron is in the +3 oxidation state.
- Iron Ore Processing: Hematite (Fe₂O₃) is a major iron ore, and its processing involves reducing iron from +3 to 0.
- Catalysis: Fe₂O₃ is used as a catalyst in various chemical reactions, where its oxidation state plays a crucial role.
Common Questions About Oxidation Numbers
What is the oxidation number of oxygen in Fe₂O₃?
Oxygen typically has an oxidation number of -2 in most compounds, including Fe₂O₃.
Can iron have other oxidation numbers?
Yes, iron can have oxidation numbers of +2, +3, and even +6 in some compounds.
How do you know if a compound is ionic or covalent?
Compounds formed between metals and non-metals are usually ionic, while those between non-metals are typically covalent. Fe₂O₃ is considered ionic due to the metal (iron) and non-metal (oxygen) combination.
Conclusion
The oxidation number of iron in Fe₂O₃ is +3, a result of the compound's structure and the typical oxidation state of oxygen. Understanding this concept is fundamental for students and professionals in chemistry, as it underpins many chemical reactions and processes. By mastering the rules for assigning oxidation numbers, you can predict the behavior of compounds and their role in various applications, from industrial processes to everyday phenomena like rust formation.
The oxidation number of iron in Fe₂O₃ is +3, a result of the compound's structure and the typical oxidation state of oxygen. Understanding this concept is fundamental for students and professionals in chemistry, as it underpins many chemical reactions and processes. By mastering the rules for assigning oxidation numbers, you can predict the behavior of compounds and their role in various applications, from industrial processes to everyday phenomena like rust formation. Whether you're studying redox reactions, analyzing material properties, or exploring the chemistry of corrosion, knowing the oxidation state of iron in Fe₂O₃ provides a solid foundation for deeper insights into the behavior of this versatile element.
The interplay of elements and states reveals deeper truths, shaping advancements across disciplines. Such insights remain pivotal in sustaining progress.
The oxidation number of iron in Fe₂O₃ is +3, a result of the compound's structure and the typical oxidation state of oxygen. Understanding this concept is fundamental for students and practitioners alike. By grasping these principles, one gains the ability to navigate chemical systems with precision, bridging theory and application. Such knowledge continues to evolve, reflecting the dynamic nature of scientific inquiry. In essence, mastery lies in recognizing how these relationships govern material properties and reactions. Thus, it stands as a testament to chemistry's pervasive influence.
The oxidation number of iron in Fe₂O₃ is +3, a result of the compound's structure and the typical oxidation state of oxygen. Understanding this concept is fundamental for students and professionals in chemistry, as it underpins many chemical reactions and processes. By mastering the rules for assigning oxidation numbers, you can predict the behavior of compounds and their role in various applications, from industrial processes to everyday phenomena like rust formation.
Whether you're studying redox reactions, analyzing material properties, or exploring the chemistry of corrosion, knowing the oxidation state of iron in Fe₂O₃ provides a solid foundation for deeper insights into the behavior of this versatile element. The interplay of elements and states reveals deeper truths, shaping advancements across disciplines. Such insights remain pivotal in sustaining progress.
The oxidation number of iron in Fe₂O₃ is +3, a result of the compound's structure and the typical oxidation state of oxygen. Understanding this concept is fundamental for students and practitioners alike. By grasping these principles, one gains the ability to navigate chemical systems with precision, bridging theory and application. Such knowledge continues to evolve, reflecting the dynamic nature of scientific inquiry. In essence, mastery lies in recognizing how these relationships govern material properties and reactions. Thus, it stands as a testament to chemistry's pervasive influence.
Latest Posts
Latest Posts
-
Light Independent Reactions Occur In The
Mar 27, 2026
-
Is A 3 2 A Good Gpa
Mar 27, 2026
-
Changing From A Liquid To A Gas
Mar 27, 2026
-
How To Make An Electric Circuit With A Switch
Mar 27, 2026
-
San Diego State University Out Of State Acceptance Rate
Mar 27, 2026