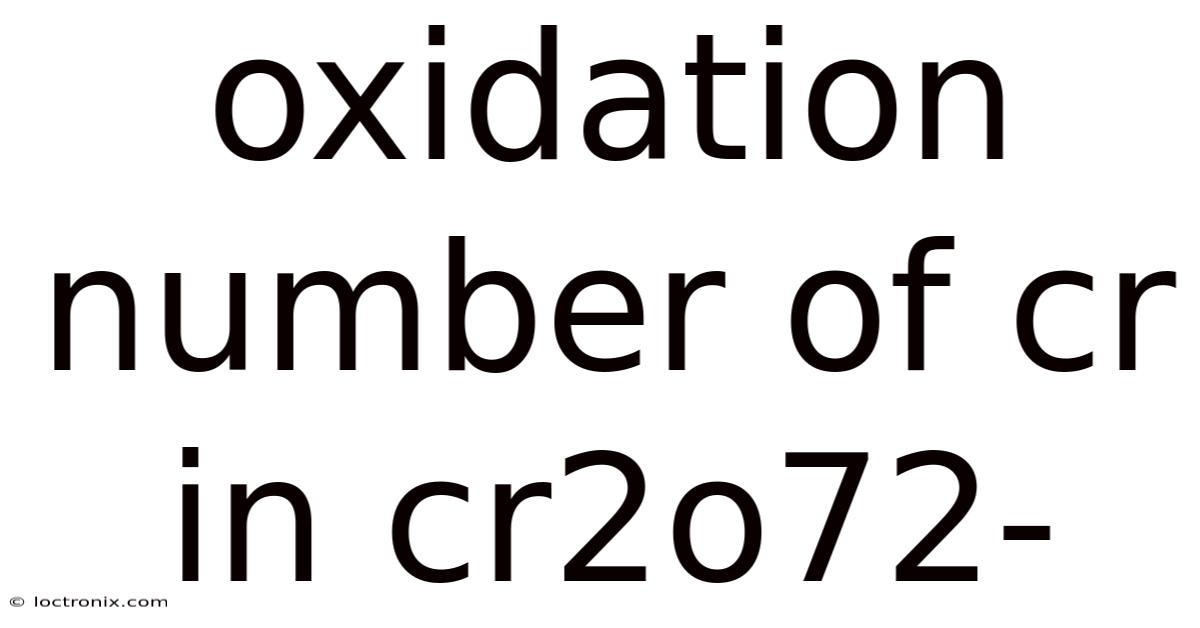The Oxidation Number of Chromium in Cr₂O₇²⁻: A practical guide
The oxidation number of chromium in the dichromate ion (Cr₂O₇²⁻) is a fundamental concept in inorganic chemistry, particularly in understanding redox reactions and the behavior of transition metals. For the dichromate ion, determining the oxidation number of chromium (Cr) is essential for analyzing its chemical properties, reactivity, and role in various chemical processes. Oxidation numbers, also known as oxidation states, represent the hypothetical charge an atom would have if all bonds to atoms of different elements were completely ionic. This article breaks down the calculation of the oxidation number of Cr in Cr₂O₇²⁻, explains the underlying principles, and highlights its significance in chemistry Simple, but easy to overlook. Practical, not theoretical..
Understanding Oxidation Numbers
Oxidation numbers are not actual charges but rather a bookkeeping tool to track electron transfer in chemical reactions. They help chemists identify which species are oxidized (lose electrons) and which are reduced (gain electrons) in redox reactions. The rules for assigning oxidation numbers are based on the assumption that all bonds are ionic, even though
TheRules That Govern Oxidation Numbers
Before we can assign a specific value to chromium in dichromate, it is useful to recall the set of conventions that chemists employ when determining oxidation numbers:
- Elements in their elemental form always have an oxidation number of 0 (e.g., Cr in metallic chromium, O₂ in dioxygen).
- The oxidation number of oxygen is usually –2, except when it is bound to fluorine (‑1) or when it is part of a peroxide (‑1) or superoxide (‑½) species.
- Hydrogen is assigned +1 when bonded to non‑metals and –1 when bonded to metals.
- The sum of oxidation numbers in a neutral compound is zero; in an ion, the sum equals the overall charge of that ion.
- The oxidation number of a monatomic ion is equal to its charge (e.g., Cl⁻ has an oxidation number of –1).
- When multiple atoms of the same element are present, the oxidation numbers are usually equal, unless the environment forces them to differ (as in certain coordination complexes).
These rules provide a systematic pathway for solving oxidation‑state problems, and they are especially straightforward when applied to polyatomic ions like dichromate.
Calculating the Oxidation Number of Chromium in Cr₂O₇²⁻
Let us now apply the rules step‑by‑step to the dichromate ion, whose formula is Cr₂O₇²⁻.
-
Identify the known oxidation numbers. - Each oxygen atom contributes –2 to the total oxidation sum.
- There are seven oxygen atoms, giving a cumulative contribution of 7 × (–2) = –14.
-
Express the unknown oxidation numbers.
- Let the oxidation number of each chromium atom be x.
- Because there are two chromium atoms, their combined contribution is 2x.
-
Write the charge‑balance equation.
[ 2x + (–14) = –2 ] The left‑hand side represents the sum of the oxidation numbers of all atoms, and it must equal the overall charge of the ion (–2). -
Solve for x.
[ 2x = –2 + 14 = 12 \quad\Rightarrow\quad x = \frac{12}{2} = +6 ]
Thus, each chromium atom in the dichromate ion carries an oxidation number of +6. This value is consistent with the oxidation state of chromium in many of its common compounds, such as chromic acid (H₂CrO₄) and chromium(VI) oxide (CrO₃) It's one of those things that adds up..
Why the +6 State Matters
The +6 oxidation state is the highest stable oxidation number exhibited by chromium, and it endows the dichromate ion with a powerful oxidizing capability. In redox chemistry, dichromate readily accepts electrons, being reduced to Cr³⁺ (oxidation number +3) in acidic solution:
[ \mathrm{Cr_2O_7^{2-} + 14,H^+ + 6,e^- \rightarrow 2,Cr^{3+} + 7,H_2O} ]
This half‑reaction illustrates how the oxidation number of chromium drops from +6 to +3, releasing six electrons in the process. Practically speaking, the ease with which dichromate can undergo this reduction underlies its widespread use as a titrant in analytical chemistry (e. g., determination of iron(II) in redox titrations) and as a cleaning agent in the semiconductor industry.
Honestly, this part trips people up more than it should It's one of those things that adds up..
Beyond redox processes, the +6 state influences the structural and electronic attributes of dichromate. The strong Cr–O bonds, the resonance‑delocalized nature of the ion, and its tetrahedral coordination geometry all stem from the high oxidation state, which draws electron density toward the oxygen atoms and stabilizes the overall framework.
This is the bit that actually matters in practice Not complicated — just consistent..
Broader Implications in Chemistry and Industry
The ability to assign and manipulate oxidation numbers is not merely an academic exercise; it is a practical tool that chemists take advantage of across disciplines:
- Catalysis: Many catalytic cycles involve changes in oxidation state, and understanding the oxidation state of a metal center helps predict reaction pathways and design more efficient catalysts.
- Materials science: Transition‑metal oxides with high oxidation states, such as Cr₂O₇²⁻‑derived compounds, serve as pigments, corrosion inhibitors, and components of electrochromic devices.
- Environmental chemistry: The redox behavior of chromium dictates its mobility and toxicity in natural waters. Cr(VI) species like dichromate are carcinogenic, whereas Cr(III) is relatively benign, making oxidation‑
The environmental chemistry point can be completed as follows:
making oxidation state control critical. Cr(VI) species, including dichromate, are highly soluble, mobile, and carcinogenic, posing significant risks to ecosystems and human health. In contrast, Cr(III) precipitates as insoluble hydroxides or oxides, effectively immobilizing it. This stark contrast drives regulatory policies and remediation strategies, such as reducing Cr(VI) to Cr(III) using reducing agents like Fe²⁺ or SO₃²⁻ before waste disposal Not complicated — just consistent..
Industrial Applications and Safety
The unique properties of Cr(VI) in dichromate enable diverse industrial roles:
- Pigments and Coatings: Chromates (e.g., PbCrO₄, "chrome yellow") provide vibrant, corrosion-resistant pigments.
- Electroplating: Chromium(VI) baths deposit hard, wear-resistant layers on metals.
- Wood Preservation: Chromated copper arsenate (CCA) leverages Cr(VI)'s biocidal action, though its toxicity has led to restricted use.
That said, these applications demand rigorous safety protocols. Modern industries increasingly seek non-Cr(VI) alternatives (e.Exposure to Cr(VI) dust or fumes causes severe health issues, including lung cancer and ulcers. g., trivalent chromium plating) to mitigate risks while maintaining functionality.
Conclusion
The assignment of chromium’s oxidation state as +6 in the dichromate ion (Cr₂O₇²⁻) exemplifies the power of oxidation numbers in decoding chemical behavior. This high oxidation state explains dichromate’s potent oxidizing ability, its structural stability, and its important role in analytical titrations, catalysis, and materials science. Conversely, it underscores the critical balance between utility and hazard, as Cr(VI) toxicity necessitates careful environmental and industrial management. At the end of the day, mastering oxidation states transcends theoretical calculation—it is a cornerstone of applied chemistry, enabling innovation while safeguarding health and the environment. As chemistry evolves, the principles governing oxidation states remain indispensable for designing sustainable processes, predicting reactivity, and harnessing the full potential of elements like chromium.