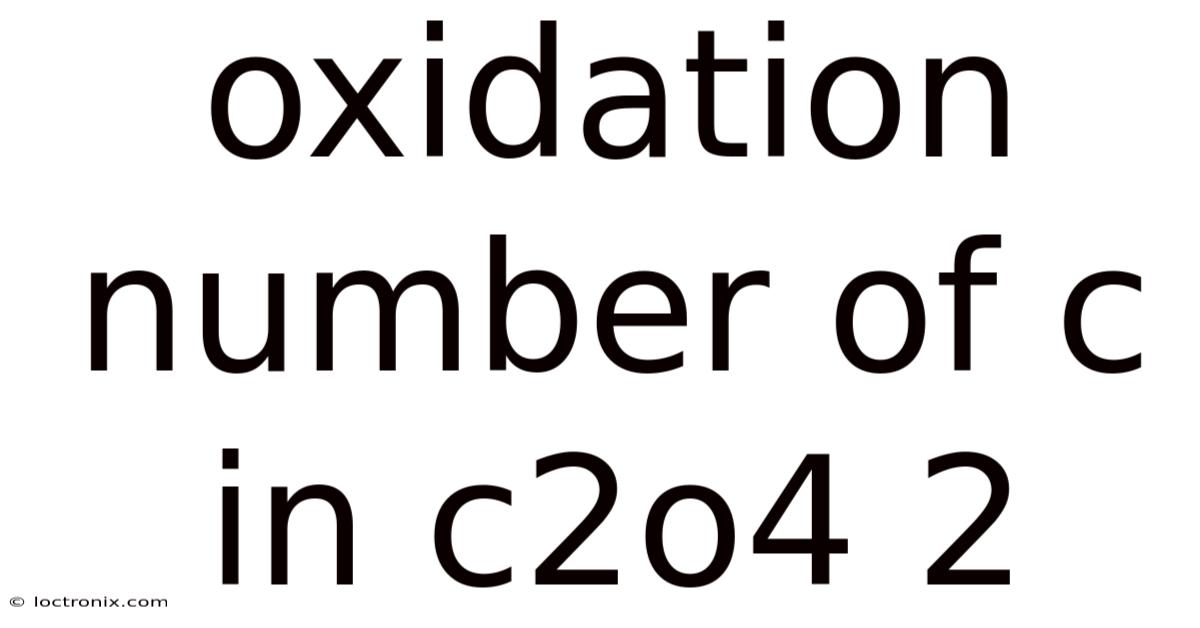The oxidation number of carbon in the oxalate ion (C₂O₄²⁻) is a fundamental concept in chemistry that helps us understand the behavior of this important polyatomic ion. Oxalate is a dianion, meaning it carries a -2 charge, and it plays a significant role in various chemical reactions, including those in biological systems and industrial processes. To determine the oxidation number of carbon in C₂O₄²⁻, we need to apply the rules of oxidation states and consider the overall charge of the ion.
First, let's recall that oxygen typically has an oxidation number of -2 in most compounds, except in peroxides where it is -1. Because of this, each oxygen atom in C₂O₄²⁻ has an oxidation number of -2. In the oxalate ion, each oxygen atom is bonded to carbon, and there are no peroxide bonds present. Since there are four oxygen atoms, the total contribution from oxygen is 4 × (-2) = -8.
You'll probably want to bookmark this section.
The oxalate ion has an overall charge of -2. In real terms, to balance this charge, the two carbon atoms must collectively have an oxidation number that, when added to the total from oxygen, equals -2. Let's denote the oxidation number of each carbon atom as x. Since there are two carbon atoms, their combined oxidation number is 2x That alone is useful..
2x + (-8) = -2
Solving for x:
2x - 8 = -2 2x = -2 + 8 2x = 6 x = 3
Which means, the oxidation number of each carbon atom in the oxalate ion (C₂O₄²⁻) is +3 Surprisingly effective..
Understanding the oxidation number of carbon in oxalate is crucial for several reasons. Take this case: in redox reactions, the oxidation state of carbon can change, indicating the transfer of electrons. In the case of oxalate, it can act as a reducing agent, donating electrons and getting oxidized to carbon dioxide (CO₂), where the oxidation number of carbon is +4. This property makes oxalate useful in various chemical processes, such as in the extraction of metals and in analytical chemistry Simple as that..
On top of that, the oxidation number of carbon in oxalate is relevant in biological systems. Oxalate is found in many plants and can form insoluble salts with calcium, leading to the formation of kidney stones in humans. The oxidation state of carbon in oxalate influences its reactivity and its ability to bind with other molecules, which is essential for understanding its role in metabolism and its potential health impacts.
All in all, the oxidation number of carbon in the oxalate ion (C₂O₄²⁻) is +3. Consider this: this value is determined by considering the oxidation states of oxygen and the overall charge of the ion. Understanding this concept is not only important for academic purposes but also for practical applications in chemistry, biology, and medicine. By grasping the oxidation states of atoms in polyatomic ions like oxalate, we can better predict and explain their chemical behavior and interactions Practical, not theoretical..
Beyond its role in biological systems, the understanding of carbon’s oxidation state in oxalate also finds significant application within industrial processes. Specifically, oxalate salts are utilized as a complexing agent in various metal extraction techniques, capitalizing on the ability of the carbon’s +3 oxidation state to form stable coordination complexes with metal ions. Day to day, this allows for the selective separation and purification of metals from ores and industrial waste streams – a process vital for producing numerous materials, from pigments to catalysts. To build on this, oxalate derivatives are employed as intermediates in the synthesis of organic compounds, leveraging the controlled oxidation and reduction of the carbon center to build complex molecular structures.
The stability of the +3 oxidation state in oxalate is intrinsically linked to its resonance structures, which distribute the double bond character across the carbon-oxygen bonds, effectively stabilizing the molecule. This resonance contributes to its relative inertness compared to other carbon compounds with similar oxidation states, making it a reliable reagent in many chemical transformations. Researchers continue to explore novel applications of oxalate, including its potential as a green solvent and as a component in advanced battery technologies, driven by its electrochemical properties and ability to participate in redox reactions The details matter here..
Finally, the consistent and predictable nature of carbon’s oxidation state in oxalate – a +3 value – provides a valuable benchmark for studying and predicting the behavior of similar compounds. On the flip side, it reinforces the fundamental principles of chemical bonding and redox chemistry, offering a tangible example of how seemingly abstract concepts translate into real-world applications. Which means, a thorough comprehension of the oxidation number of carbon in C₂O₄²⁻ serves as a cornerstone for deeper explorations into the intricacies of chemical reactivity and its diverse manifestations across numerous scientific disciplines.
The +3 oxidation state of carbon in the oxalate ion (C₂O₄²⁻) not only underpins its structural stability but also dictates its reactivity in chemical systems. On top of that, for instance, in homogeneous catalysis, oxalate-based ligands can stabilize metal centers, enhancing their activity in reactions such as oxidation or hydrogenation. This oxidation state enables oxalate to act as a versatile ligand in coordination chemistry, where it forms complexes with transition metals. These complexes are central in catalysis, as the delocalized electron density from the resonance structures allows for efficient electron transfer processes. This property is particularly valuable in industrial settings where precise control over reaction conditions is critical It's one of those things that adds up..
Beyond traditional applications, the unique characteristics of the oxalate ion are driving innovation in sustainable technologies. Consider this: in the realm of green chemistry, oxalate derivatives are being explored as eco-friendly solvents due to their low toxicity and ability to dissolve a wide range of organic and inorganic compounds. Their use in solvent systems reduces reliance on volatile organic compounds, aligning with global efforts to minimize environmental impact. Additionally, the redox-active nature of the +3 oxidation state is being harnessed in energy storage solutions Not complicated — just consistent..
Not the most exciting part, but easily the most useful The details matter here..
a promising alternative to conventional battery technologies. Beyond that, the inherent stability of the oxalate ion and its ability to undergo controlled decomposition pathways are being investigated for applications in biodegradable polymers and controlled-release drug delivery systems. The potential to tailor the degradation rate of these materials by manipulating the oxalate structure opens exciting avenues for creating environmentally benign and precisely timed release mechanisms Less friction, more output..
The study of oxalate’s oxidation state extends beyond simple quantification; it’s a window into understanding the interplay of electronic structure and chemical behavior. These methods allow researchers to predict and rationalize the observed reactivity, guiding the design of new oxalate-based materials with tailored properties. Computational modeling and spectroscopic techniques, such as X-ray diffraction and electron paramagnetic resonance, provide detailed insights into the bonding arrangements and electronic transitions within the oxalate ion and its complexes. The relatively simple yet reliable structure of C₂O₄²⁻ provides an ideal platform for testing theoretical models and refining our understanding of fundamental chemical principles.
Pulling it all together, the consistent +3 oxidation state of carbon within the oxalate ion is far more than a mere numerical value. It represents a cornerstone of its chemical identity, dictating its structural integrity, reactivity, and ultimately, its diverse applications. In practice, from its established role as a ligand in catalysis to its burgeoning potential in sustainable technologies, the oxalate ion exemplifies how a carefully defined oxidation state can get to a wealth of possibilities across chemistry, materials science, and energy research. Continued investigation into the intricacies of this seemingly simple compound promises to yield further breakthroughs and solidify its position as a valuable tool for scientific advancement.