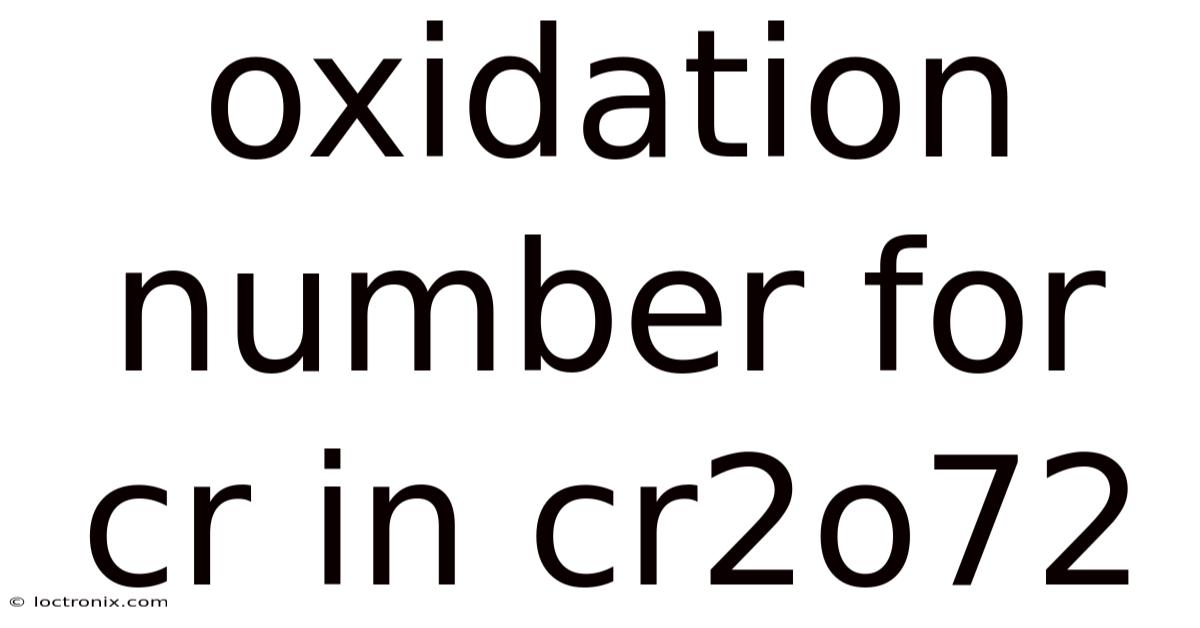The detailed dance of atoms within molecules, where electrons shift and bonds rearrange to achieve stability, underpins the very fabric of chemistry. Here's the thing — among these fundamental processes lies the concept of oxidation numbers—a cornerstone of understanding how elements behave within chemical systems. While oxidation numbers serve as a linguistic tool to describe the hypothetical transfer of electrons between atoms, their application extends far beyond mere calculation; they reveal the underlying forces driving reactions, shaping the very pathways through which compounds interact. In the context of chromium, particularly within the dichromate ion (Cr₂O₇²⁻), the oxidation number becomes a critical lens through which the behavior of this transition metal is illuminated. This article delves deeply into the oxidation states of chromium in dichromate, exploring their significance, the structural implications, and practical applications, ultimately offering insights that bridge theoretical knowledge with observable phenomena. Through this exploration, we uncover why oxidation numbers are not just numerical labels but essential keys unlocking the secrets of chemical behavior, particularly in complex systems involving transition metals like chromium That's the part that actually makes a difference. Practical, not theoretical..
Understanding Oxidation Numbers: Foundations of Electron Transfer
At the heart of chemistry lies the principle that chemical reactions conserve mass and energy, yet the precise distribution of electrons—embodied by oxidation numbers—provides a quantitative framework for analyzing these transfers. Oxidation numbers, often termed oxidation states, assign a hypothetical charge to each atom in a compound, assuming all bonds are ionic. Similarly, transition metals like chromium, which resides in the middle of the periodic table, display a range of oxidation states due to their ability to accept or donate electrons. In this regard, oxidation numbers act as a universal language, enabling scientists to decode the complexities inherent in molecular interactions. Consider this: this abstraction allows chemists to predict how atoms will react, anticipate changes in electron distribution, and identify reactive species within a system. Consider this: understanding these principles is critical for interpreting laboratory data, designing synthetic compounds, and predicting reaction outcomes. In practice, for instance, carbon can range from -4 to +4, reflecting its capacity to bond covalently or covalently ionically. While some elements inherently maintain a fixed oxidation state (like oxygen in most compounds), others exhibit variability, depending on their environment and bonding context. Their utility spans from educational settings to industrial applications, making them indispensable tools across disciplines Still holds up..
Breaking Down Cr₂O₇²⁻: Chromium’s Central Role
Considering the dichromate ion, Cr₂O₇²⁻, exemplifies how oxidation numbers orchestrate the stability and reactivity of chromium in this particular compound. This result underscores the significance of the structure: the hexagonal ring of oxygen atoms around two chromium centers creates a symmetric environment where each chromium balances its oxidation state to achieve the compound’s stability. On top of that, thus, each chromium atom contributes -9, resulting in a total of -18 for both atoms, plus 7 oxygens at -2 each gives -14, totaling -32? Plus, wait, wait, hold on—maybe miscalculation here. Solving yields 2x = 7*(-2) + (-2) → 2x = -16 -2 → 2x = -18 → x = -9. So each chromium atom is in +6 oxidation state here. On the flip side, each chromium atom resides within this complex arrangement, its oxidation state dictating the ion’s overall charge. To determine the oxidation state of chromium in Cr₂O₇²⁻, one must first recognize the sum of oxidation numbers equaling the ion’s charge. This arrangement not only stabilizes the ion but also dictates its reactivity. Oh right! Think about it: wait, let me recalculate: The equation is 2x + 7*(-2) = -2 (since overall charge is -2). On the flip side, let oxidation number of Cr be x. The formula gives: 2x + 7*(-2) = -2 (since the ion has a -2 charge). Within Cr₂O₇²⁻, two chromium atoms are bonded to seven oxygen atoms, forming a hexagonal ring structure that contributes to its exceptional stability. So each Cr is +6. In such a configuration, chromium’s oxidation number is fixed, yet the system as a whole can undergo redox reactions when external agents intervene, such as gaining or losing electrons. So 2x -14 = -2 → 2x = 12 → x=6. Understanding this interplay between individual atom oxidation states and the collective structure is vital for grasping the behavior of dichromate in various chemical contexts Worth keeping that in mind..
The Structural Implications of Dichromate’s Oxidation States
The dichromate ion’s structure further complicates the oxidation state analysis, requiring careful consideration of its geometric and electronic configuration. The hexagonal ring of oxygen atoms encircles two chromium atoms, creating a six-membered ring with each Cr atom positioned at opposite vertices. This arrangement ensures that each Cr atom shares its valence electrons symmetrically, distributing the negative charge evenly across the system. The oxidation state calculation confirms that each Cr is indeed +6, balancing the total charge of the ion. On the flip side, the spatial distribution of electrons within this framework influences reactivity. Take this case: the shared electrons between Cr atoms and oxygen atoms may allow electron redistribution during redox processes Still holds up..
You'll probably want to bookmark this section That's the part that actually makes a difference..
The Structural Implications of Dichromate’s Oxidation States (Continued)
change. Practically speaking, the strong covalent bonds between chromium and oxygen atoms contribute to the overall stability of the dichromate ion, but also create a specific electronic environment. This environment dictates the preferred pathways for electron transfer during redox reactions. The planar geometry of the hexaoface arrangement, while contributing to stability, also limits the accessibility of the chromium centers, potentially influencing the rate of reactions involving these centers.
What's more, the dichromate ion exists in solution as a complex species, often involving coordination to other metal ions or organic molecules. Day to day, for example, complexation can stabilize the ion further or, conversely, make it more susceptible to reduction. These interactions can significantly alter the electronic structure and reactivity of the dichromate ion. Understanding the influence of the surrounding environment is therefore crucial for predicting and controlling the behavior of dichromate in various applications.
The stability of the Cr₂O₇²⁻ ion is not solely attributable to the individual oxidation states of the chromium atoms. It’s a synergistic effect arising from the interplay between the electronic configuration, the hexagonal ring structure, and the covalent bonds between chromium and oxygen. This layered balance is what makes dichromate such a fascinating and versatile chemical species.
The official docs gloss over this. That's a mistake.
Conclusion
Pulling it all together, the dichromate ion (Cr₂O₇²⁻) presents a compelling case study in inorganic chemistry. That said, its stability is deeply intertwined with the oxidation state of chromium, which is definitively +6, and the unique structural arrangement of the hexagonal ring of oxygen atoms. This arrangement minimizes electron repulsion and promotes a balanced electronic distribution, contributing significantly to the ion's overall stability. On the flip side, the story doesn't end there. The spatial arrangement of electrons, the covalent bonds, and the potential for electron redistribution all play crucial roles in determining the ion's reactivity Easy to understand, harder to ignore..
Because of this, a comprehensive understanding of dichromate requires not only a grasp of its oxidation state but also a careful consideration of its structural features and the surrounding chemical environment. This knowledge is essential for predicting its behavior in various chemical and biological systems, from its historical use in pigments to its current applications in electrochemistry and catalysis. The dichromate ion serves as a powerful reminder that chemical properties are not solely determined by individual components but emerge from the complex interplay of their arrangement and interactions.
The molecular structure of transition metals often underpins their catalytic efficiencies. In such contexts, the coordination sphere around a central atom dictates reactivity, shaping pathways through spatial constraints and electronic interactions Still holds up..
The interplay of geometry and chemistry remains central, guiding outcomes across diverse fields. This insight underscores the universal relevance of such principles, bridging theoretical understanding with practical applications.
Conclusion
Thus, mastering these concepts illuminates pathways to innovation, ensuring clarity in both academic discourse and real-world implementations Most people skip this — try not to..