How Many Electrons Does An Atom Of Oxygen Have
loctronix
Mar 18, 2026 · 6 min read
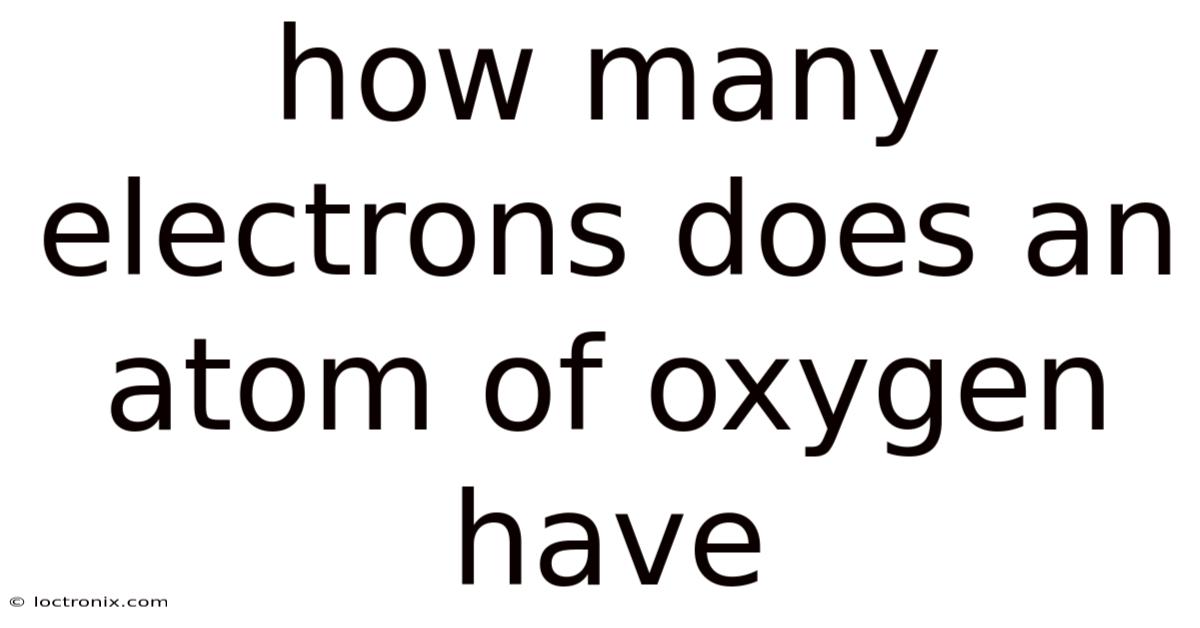
Table of Contents
Understanding the number of electrons in an atom of oxygen is a fundamental concept in chemistry that helps us grasp the basics of atomic structure. When we explore this topic, we uncover the building blocks of matter and how they interact. The answer to this question is not just a number but a key to unlocking the secrets of how elements behave and form compounds. Let’s dive into the details and discover why this information matters.
First, let’s clarify what an atom is. Atoms are the smallest units of matter that retain the properties of an element. Each atom has a nucleus at its center, which contains protons and neutrons, and surrounding electrons. The number of electrons in an atom is crucial because it determines how atoms interact with one another. For example, the number of electrons affects the chemical reactions and the formation of bonds. Understanding this helps us explain why oxygen is a vital element in life and why it plays a role in many everyday processes.
Now, let’s focus on the element oxygen. Oxygen is a chemical element with the symbol O and atomic number 8. This means it has eight protons in its nucleus. Each element has a unique number of protons, which defines its identity. The atomic number tells us the number of electrons in a neutral atom. For oxygen, this number is eight, which means a neutral oxygen atom has eight electrons. However, in most chemical reactions, atoms gain or lose electrons to achieve stability. This is where the concept of oxidation comes into play, and it’s essential to understand how this affects the electron count.
To determine the number of electrons in an oxygen atom, we need to consider its position in the periodic table. Oxygen is in the second period and the fifth group. Its electron configuration is 1s² 2s² 2p⁴. This means the outermost shell of oxygen has four electrons. But wait—this is a bit confusing. Let’s break it down carefully.
The atomic structure of oxygen involves electrons filling different energy levels. The first two shells contain two electrons each, and the third shell holds four. However, the total number of electrons in a neutral atom is the sum of all these. For oxygen, the total number of electrons is 8. This is because the atomic number (number of protons) is 8, and for a neutral atom, the number of electrons equals the number of protons. So, oxygen has eight electrons in its neutral state.
But why is this important? Because the number of electrons directly influences how atoms bond with each other. For instance, when oxygen combines with another element, it often gains or loses electrons to reach a stable configuration. This process is central to understanding reactions and the formation of compounds. If we know how many electrons an atom has, we can predict its behavior in different chemical scenarios.
Let’s explore the steps involved in understanding this. First, we identify the atomic structure of oxygen. The nucleus contains protons and neutrons, but the electrons are the ones we’re interested in. In a neutral atom, the number of electrons equals the number of protons. Since oxygen has eight protons, it must have eight electrons. However, in real-world scenarios, atoms often change their electron count. For example, when oxygen forms a bond, it may gain or lose electrons to achieve a stable electron configuration. This is where the concept of valence electrons comes into play.
Valence electrons are the electrons in the outermost shell. For oxygen, these are the two electrons in the second energy level. But let’s clarify this with a clearer breakdown. Oxygen’s electron configuration is 1s² 2s² 2p⁴. The two electrons in the 2p shell are the valence electrons. This means that when oxygen forms bonds, it typically uses these valence electrons to connect with other atoms.
Now, let’s move to the steps involved in calculating the number of electrons. When we talk about the number of electrons in an atom, we’re referring to the total number of electrons present. For oxygen, this is straightforward: it has eight electrons. However, in some contexts, we might refer to the atomic number and the electron configuration. The atomic number is the number of protons, which is 8 for oxygen. The electron configuration tells us how these protons are arranged.
It’s important to note that while the atomic number gives us the number of protons, the actual number of electrons in a neutral atom is determined by the number of electrons in the outermost shell. For oxygen, this is eight. But when it comes to chemical reactions, the actual number of electrons can vary. For example, in water (H₂O), oxygen has six electrons in its outer shell. This shows how the number of electrons can change during bonding.
Understanding the number of electrons in an atom helps us grasp the concept of chemical bonding. Atoms tend to gain, lose, or share electrons to achieve a stable electron configuration. This is the foundation of chemistry. For oxygen, its ability to form bonds is closely tied to its electron count. If we know how many electrons it has, we can predict its interactions with other elements.
Now, let’s address some common questions that arise when discussing this topic. What if an atom has more or fewer electrons? This is a great point. If an atom has fewer electrons than its atomic number, it becomes unstable and may react to gain electrons. Conversely, if it has more, it might lose electrons. This explains why elements like oxygen are reactive. The balance of electrons is what makes chemistry so fascinating.
Another important aspect is the symbol* and definition of electrons. In chemistry, we often refer to electrons as the particles that carry electric charge. The number of electrons in an atom is what defines its chemical properties. For example, oxygen’s high electronegativity is due to its tendency to attract electrons. This makes it a key player in many chemical reactions.
To reinforce this understanding, let’s look at the scientific explanation behind this. The electron shell model helps us visualize how electrons are arranged. Each shell can hold a certain number of electrons, and the outermost shell determines the chemical behavior. For oxygen, the second shell has four electrons, which is why it often forms compounds by sharing or transferring electrons.
In conclusion, the number of electrons in an atom of oxygen is eight. This might seem simple, but it’s a crucial detail that shapes how we understand the element’s role in the world. Whether you’re studying chemistry, preparing for exams, or just curious about the building blocks of matter, knowing this fact is essential. It connects us to the invisible forces that govern everything from living organisms to the air we breathe.
By exploring this topic, we not only learn about the basics of atomic structure but also appreciate the complexity of the elements that make up our universe. This knowledge empowers us to make informed decisions in science, technology, and everyday life. So, the next time you think about oxygen, remember its electron count and the significance it holds in the grand scheme of things.
Latest Posts
Latest Posts
-
How To Type Less Than Or Equal To
Mar 18, 2026
-
Thousand Million Billion Trillion Quadrillion Quintillion Sextillion
Mar 18, 2026
-
What Two Factors Does Air Pressure Depend On
Mar 18, 2026
-
What Are The Five Outer Planets
Mar 18, 2026
-
Long Vowel And Short Vowel Rules
Mar 18, 2026
Related Post
Thank you for visiting our website which covers about How Many Electrons Does An Atom Of Oxygen Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.