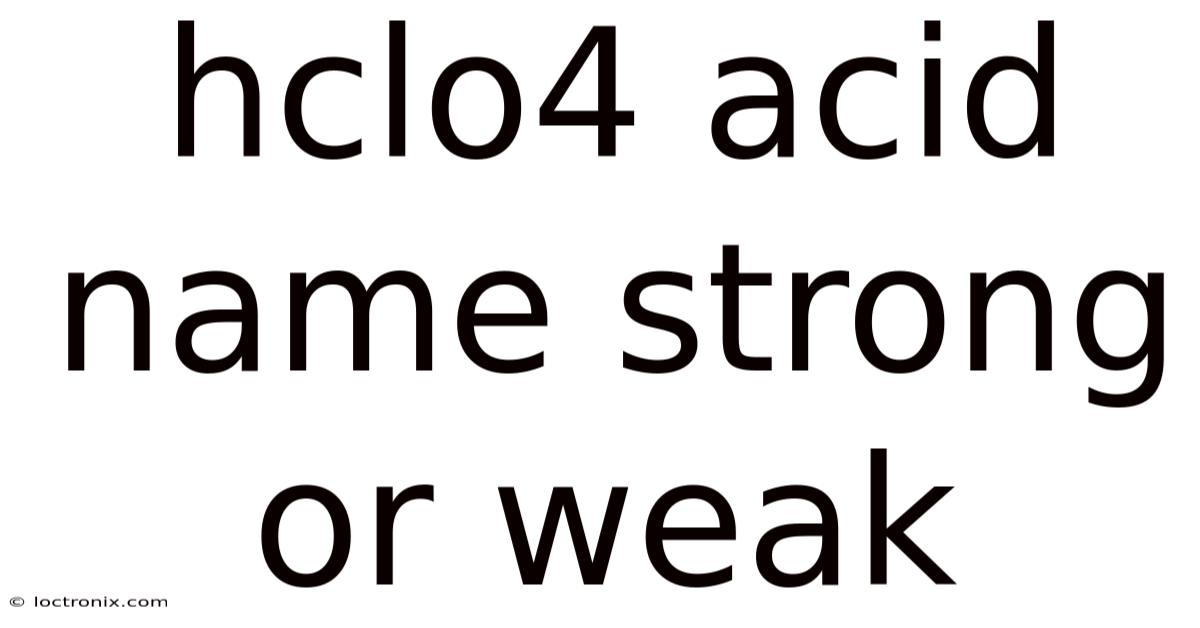Understanding the properties and significance of HClO4 acid is essential for students and professionals alike. In real terms, this powerful chemical compound, known for its strong acidity, plays a vital role in various scientific and industrial applications. In this article, we will dig into the details of HClO4 acid, exploring its name, strength, and the implications of its chemical behavior. By the end of this discussion, you will have a clearer picture of why this acid is considered one of the most potent in the world of chemistry And that's really what it comes down to..
HClO4, or perchloric acid, is a highly reactive and strong acid that has garnered significant attention due to its unique characteristics. This structure contributes to its remarkable acidity, making it a key player in both laboratory settings and industrial processes. Worth adding: its molecular formula, HClO4, indicates that it contains four chlorine atoms bonded to four hydrogen atoms and one oxygen atom. When HClO4 reacts with water, it forms hydrochloric acid and oxygen gas, a reaction that highlights its strength. This transformation is crucial for understanding its behavior in different environments and applications Not complicated — just consistent..
The strength of HClO4 is not just a matter of its chemical formula; it also influences how it interacts with other substances. As a strong acid, HClO4 dissociates completely in water, releasing a high concentration of hydrogen ions. Day to day, this property allows it to effectively lower the pH of solutions, making it an essential component in various chemical reactions. The ability of HClO4 to donate protons is what sets it apart from weaker acids, which only partially dissociate. This distinction is vital for students studying acid-base chemistry, as it emphasizes the importance of understanding acid strength in scientific contexts Which is the point..
In educational settings, it is crucial to grasp the implications of HClO4's strength. Also, its high acidity can pose safety risks if not handled properly. And students must be aware of the potential hazards associated with this compound, including its ability to react violently with organic materials. This knowledge is not just academic; it has real-world applications in fields such as environmental science and chemical manufacturing. By recognizing the dangers of strong acids like HClO4, learners can appreciate the importance of safety protocols in the laboratory and industry That alone is useful..
The significance of HClO4 extends beyond its chemical properties. This connection between chemistry and environmental health underscores the need for responsible handling and disposal of such substances. In the realm of environmental science, understanding the behavior of strong acids is essential for addressing pollution and contamination issues. Take this: when HClO4 is released into water bodies, it can disrupt ecosystems and affect aquatic life. Students should consider how their understanding of HClO4 contributes to broader discussions about sustainability and chemical safety.
When exploring the properties of HClO4, it is the kind of thing that makes a real difference. The acid is commonly used in the production of fertilizers, where it helps to enhance the availability of nutrients for plants. Day to day, additionally, HClO4 is employed in the manufacturing of certain chemicals and pharmaceuticals, showcasing its versatility. These applications highlight the practical importance of understanding HClO4's strength and reactivity. By learning about these uses, students can appreciate the relevance of chemistry in everyday life and the potential for innovation.
Worth adding, the study of HClO4 provides a valuable opportunity for students to engage with fundamental concepts in chemistry. Understanding the principles of acid strength helps in grasping more complex topics, such as buffer solutions and titrations. In practice, these concepts are foundational in both academic and professional settings, emphasizing the need for a solid grasp of basic chemical principles. As learners progress in their studies, they will find that their knowledge of HClO4 enhances their ability to tackle challenging problems and concepts.
Easier said than done, but still worth knowing It's one of those things that adds up..
In the context of safety, make clear the precautions that must be taken when working with strong acids like HClO4 — this one isn't optional. Proper ventilation, protective equipment, and careful handling are critical to ensuring safety in the laboratory. Students should be aware of the potential for fire and explosion hazards when mixing HClO4 with other substances. By prioritizing safety, learners not only protect themselves but also contribute to a culture of responsibility in scientific practices That's the part that actually makes a difference..
Some disagree here. Fair enough.
The scientific explanation of HClO4's behavior also invites deeper inquiry into its reactivity. And when it comes into contact with bases, HClO4 reacts vigorously, often producing heat and potentially dangerous gases. Plus, this reactivity is a key factor in its classification as a strong acid. Practically speaking, understanding these reactions can help students develop critical thinking skills, as they analyze how different substances interact. This process encourages a more nuanced understanding of chemical interactions and the importance of controlling variables in experiments.
As we explore the topic further, it becomes clear that HClO4 is more than just a chemical compound; it is a subject that bridges various disciplines. In biology, for example, perchloric acid is sometimes used in research settings to study cellular processes. Still, its strong acidity can affect enzyme activity and cellular metabolism, making it a valuable tool in scientific investigations. This application highlights the interconnectedness of chemistry with other fields, reinforcing the importance of a comprehensive understanding of HClO4.
All in all, the discussion surrounding HClO4 acid underscores its significance in both academic and practical contexts. By delving into the details of its properties and implications, students can enhance their knowledge and develop a deeper appreciation for the role of chemistry in our world. In real terms, understanding HClO4 is not just about memorizing facts; it is about building a foundation for future learning and innovation. Worth adding: its strong acidity, coupled with its versatile applications, makes it a compound that deserves careful consideration. As we continue to explore the complexities of this powerful acid, let us remember the importance of safety, responsibility, and curiosity in our pursuit of scientific knowledge It's one of those things that adds up..