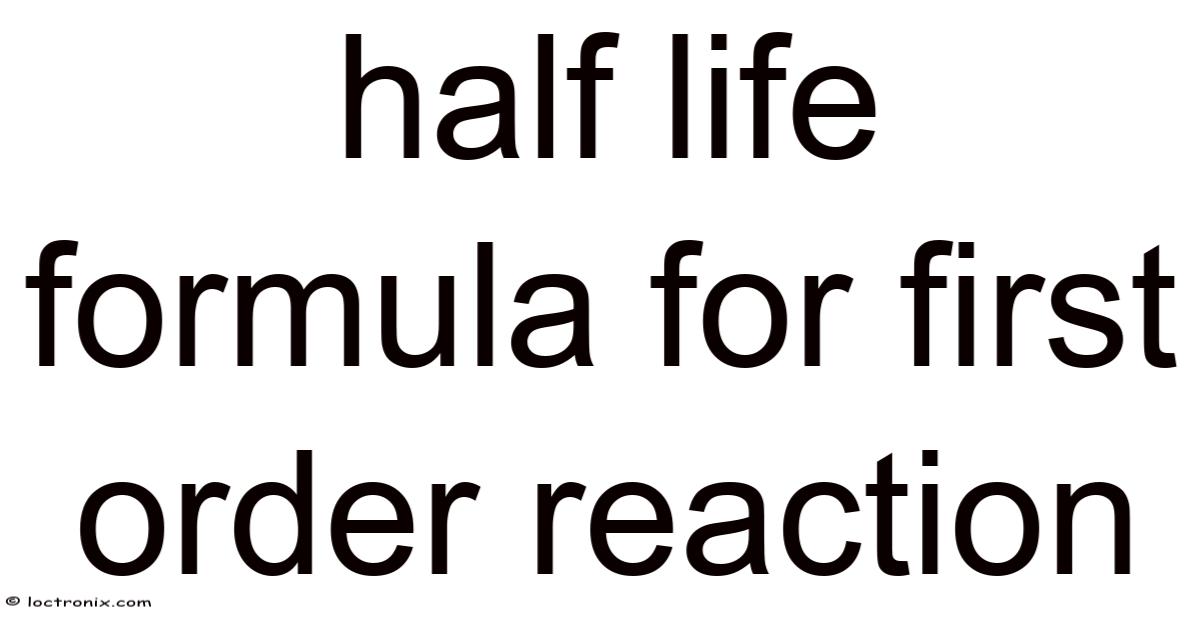Introduction: Understanding the Half‑Life Formula for a First‑Order Reaction
The half‑life (t½) of a first‑order reaction is the time required for the concentration of a reactant to decrease to half of its initial value. This simple yet powerful concept appears in fields ranging from pharmaceutical kinetics to environmental engineering, and mastering its formula provides a gateway to predicting how quickly a system evolves. In this article we will explore the derivation of the first‑order half‑life equation, examine its practical applications, discuss common pitfalls, and answer frequently asked questions—all while keeping the mathematics clear and the chemistry intuitive.
1. The Fundamentals of First‑Order Kinetics
1.1 Definition of a First‑Order Reaction
A reaction is first order when its rate depends linearly on the concentration of a single reactant:
[ \text{Rate} = -\frac{d[A]}{dt}=k[A] ]
where
- ([A]) = concentration of reactant A (mol L⁻¹)
- (k) = first‑order rate constant (s⁻¹)
- (t) = time (s)
The negative sign indicates that the reactant concentration decreases with time Simple, but easy to overlook..
1.2 Integrated Rate Law
Integrating the differential expression gives the integrated first‑order rate law:
[ \ln\frac{[A]}{[A]_0}= -kt ]
or, equivalently,
[ [A]=[A]_0 e^{-kt} ]
where ([A]_0) is the initial concentration at (t = 0). This exponential decay relationship is the cornerstone for deriving the half‑life formula Easy to understand, harder to ignore. Took long enough..
2. Deriving the Half‑Life Expression
The half‑life, (t_{½}), is defined by the condition ([A] = \frac{1}{2}[A]_0). Substituting this into the integrated law:
[ \ln\frac{\frac{1}{2}[A]_0}{[A]0}= -k t{½} ]
[ \ln\frac{1}{2}= -k t_{½} ]
Since (\ln\frac{1}{2}= -\ln 2),
[ -\ln 2 = -k t_{½} ]
[ t_{½}= \frac{\ln 2}{k} ]
Because (\ln 2 \approx 0.693), the half‑life formula for a first‑order reaction is often written as:
[ \boxed{t_{½}= \frac{0.693}{k}} ]
2.1 Key Features of the Formula
- Independence from initial concentration – Unlike higher‑order reactions, the half‑life does not contain ([A]_0). What this tells us is, regardless of how much reactant you start with, the time to halve the concentration remains constant.
- Direct proportionality to the inverse of the rate constant – Faster reactions (larger (k)) have shorter half‑lives, while slower reactions (smaller (k)) persist longer.
These characteristics make the first‑order half‑life a convenient metric for comparing kinetic data across experiments Small thing, real impact..
3. Practical Applications
3.1 Radioactive Decay
Radioisotopes follow first‑order kinetics because each nucleus decays independently. The half‑life formula enables scientists to predict how long a sample remains hazardous, design medical imaging protocols, and date archaeological finds Not complicated — just consistent..
3.2 Drug Metabolism
Many pharmaceuticals are eliminated from the bloodstream by first‑order processes (e.g., hepatic metabolism). Clinicians use the half‑life to schedule dosing intervals, ensuring therapeutic levels without accumulation And it works..
3.3 Environmental Degradation
The breakdown of pollutants such as pesticides often follows first‑order kinetics in soil or water. Knowing the half‑life helps regulators assess persistence, set safe exposure limits, and plan remediation timelines.
3.4 Chemical Engineering
In continuous‑flow reactors, the residence time distribution can be approximated by first‑order decay for certain side reactions. Engineers use the half‑life to size reactors and control product quality.
4. Step‑by‑Step Example: Calculating Half‑Life from Experimental Data
Suppose you monitor the concentration of a reactant A over time and obtain the following data:
| Time (s) | ([A]) (mol L⁻¹) |
|---|---|
| 0 | 0.100 |
| 200 | 0.Worth adding: 060 |
| 400 | 0. 036 |
| 600 | 0. |
Step 1 – Determine the rate constant (k).
Plot (\ln[A]) versus (t); the slope equals (-k). Using two points (0 s, (\ln0.100)) and (400 s, (\ln0.036)):
[ k = -\frac{\ln0.036 - \ln0.Day to day, 100}{400 \text{ s}} = -\frac{-3. 327 - (-2.303)}{400}= \frac{1.024}{400}=2.
Step 2 – Apply the half‑life formula.
[ t_{½}= \frac{0.693}{2.56\times10^{-3}\ \text{s}^{-1}} \approx 270\ \text{s} ]
Step 3 – Verify with data.
At 270 s, the concentration should be roughly 0.050 mol L⁻¹. Interpolating between the 200 s (0.060) and 400 s (0.036) points confirms the estimate, demonstrating the reliability of the formula Took long enough..
5. Common Misconceptions
| Misconception | Why It’s Incorrect | Correct Understanding |
|---|---|---|
| Half‑life depends on the initial concentration. | While generally true, temperature and catalysts can change (k) dramatically without altering the observed half‑life if the system is not at constant conditions. Plus, | |
| *A larger half‑life always means a slower reaction. Worth adding: | For first‑order reactions, (t_{½}) is constant, independent of ([A]_0). Here's the thing — * | The derivation assumes a linear rate law. Because of that, |
| *The half‑life formula works for any reaction order. | Use (t_{½}= \frac{1}{k[A]0}) for second order, or (t{½}= \frac{[A]_0}{2k}) for zero order. * | This is true for second‑order or zero‑order reactions, but not for first order. |
6. Factors Influencing the First‑Order Rate Constant
- Temperature – According to the Arrhenius equation, (k = A e^{-E_a/RT}). Raising temperature increases (k) and shortens (t_{½}).
- Catalysts – Lower the activation energy (E_a), boosting (k) and reducing half‑life.
- Solvent Effects – Polarity and dielectric constant can stabilize transition states, altering (k).
- pH (for acid/base‑catalyzed reactions) – Changes in proton concentration may switch the reaction mechanism, effectively changing the order.
Understanding these influences allows chemists to engineer half‑lives for desired outcomes, such as designing a drug with a 12‑hour dosing interval Easy to understand, harder to ignore..
7. Extending the Concept: Multiple‑Half‑Life Calculations
Because the half‑life is constant, the concentration after (n) half‑lives is simply:
[ [A] = [A]_0 \left(\frac{1}{2}\right)^n ]
| Number of Half‑Lives (n) | Remaining Fraction of ([A]_0) | Remaining % |
|---|---|---|
| 1 | (1/2) | 50 % |
| 2 | (1/4) | 25 % |
| 3 | (1/8) | 12.On top of that, 5 % |
| 4 | (1/16) | 6. 25 % |
| 5 | (1/32) | 3. |
This geometric decay makes it straightforward to estimate how long it will take for a reactant to become negligible (often taken as <5 % of the original amount, which occurs after ~4–5 half‑lives) Small thing, real impact. Still holds up..
8. Frequently Asked Questions (FAQ)
Q1: Can the half‑life be measured directly without calculating (k)?
Yes. By monitoring the concentration until it reaches half the initial value, the elapsed time is the half‑life. This experimental (t_{½}) can then be used to compute (k = 0.693/t_{½}) Worth knowing..
Q2: Does the presence of a reversible reaction affect the half‑life?
Only if the reverse reaction is comparable in rate. In a truly first‑order irreversible process, the simple formula holds. For reversible systems, the observed half‑life becomes a function of both forward and reverse rate constants.
Q3: How accurate is the 0.693 approximation?
Very accurate. (\ln 2) is exactly 0.693147..., and using 0.693 introduces a negligible error (<0.01 %). For high‑precision work, retain more digits.
Q4: Why do some textbooks present the half‑life as (t_{½}= \frac{\ln 2}{k}) while others use (\frac{0.693}{k})?
Both are mathematically identical; the latter is a convenient decimal approximation for quick calculations.
Q5: If a reaction follows mixed‑order kinetics, can I still use the first‑order half‑life formula?
Only in the region where first‑order behavior dominates. To give you an idea, a reaction may be pseudo‑first‑order when one reactant is in large excess. Verify the linearity of a (\ln[A]) vs. (t) plot before applying the formula Worth keeping that in mind..
9. Practical Tips for Laboratory Work
- Choose an appropriate detection method (spectrophotometry, chromatography) that provides a linear response to concentration.
- Collect data at early times to capture the exponential decay accurately; later points become noisy as concentrations approach detection limits.
- Plot both (\ln[A]) vs. (t) and ([A]) vs. (t). A straight line in the log plot confirms first‑order behavior.
- Maintain constant temperature using a thermostated bath; even a few degrees shift can change (k) noticeably.
- Report the half‑life with its uncertainty, derived from the standard error of the slope in the linear regression of the log plot.
10. Conclusion: Why Mastering the Half‑Life Formula Matters
The half‑life formula for a first‑order reaction, (t_{½}=0.693/k), is more than a textbook equation; it is a practical tool that links kinetic theory to real‑world decisions. Whether you are designing a drug dosing schedule, estimating the longevity of a contaminant, or simply interpreting experimental data, understanding how the half‑life emerges from the integrated rate law empowers you to predict, control, and communicate chemical change with confidence The details matter here. Which is the point..
By recognizing its derivation, appreciating its independence from initial concentration, and applying it thoughtfully across disciplines, you turn a simple mathematical expression into a versatile lens through which the temporal behavior of countless systems becomes clear. Keep the formula at hand, test its assumptions, and let the elegance of first‑order kinetics guide your scientific and engineering endeavors Nothing fancy..