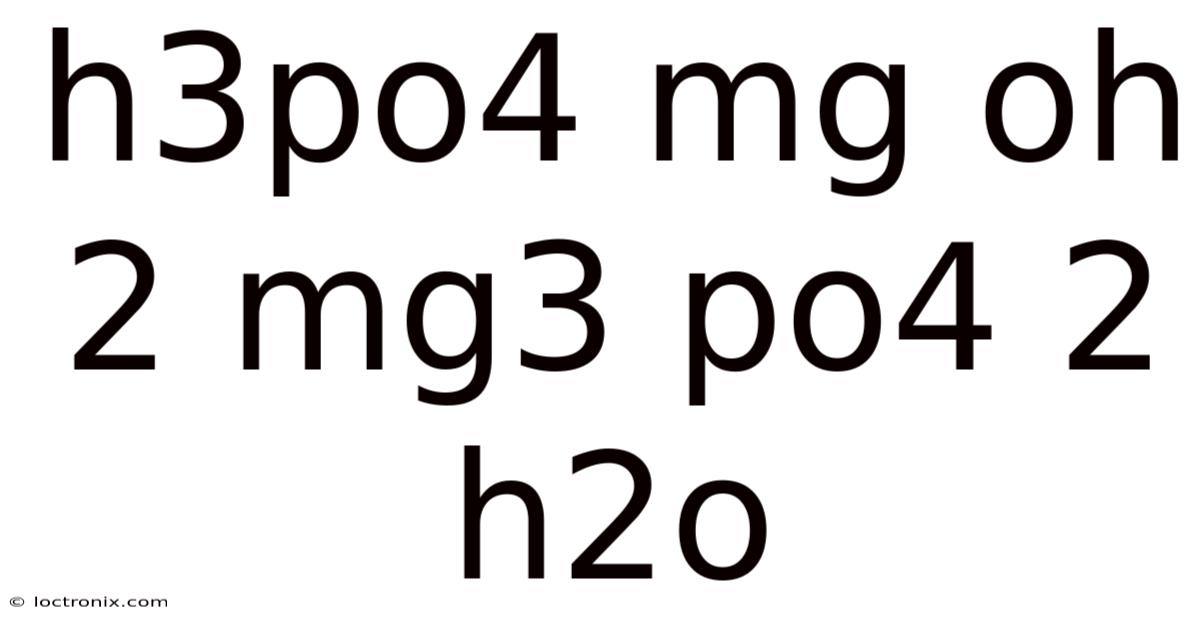H3PO4 + Mg(OH)2 → Mg3(PO4)2 + H2O: Complete Chemical Reaction Guide
The chemical reaction between phosphoric acid (H3PO4) and magnesium hydroxide (Mg(OH)2) represents an important acid-base neutralization process that produces magnesium phosphate (Mg3(PO4)2) and water (H2O). Which means this reaction is widely studied in chemistry due to its practical applications in various industrial and laboratory settings. Understanding the mechanics, balancing, and implications of this reaction provides valuable insights into inorganic chemistry principles and real-world applications.
Understanding the Reactants
Phosphoric Acid (H3PO4)
Phosphoric acid is a weak acid with the chemical formula H3PO4. This triprotic acid contains three replaceable hydrogen atoms, meaning it can donate up to three protons (H+) in acid-base reactions. But in its pure form, it appears as a colorless, odorless crystalline solid that readily dissolves in water to form a clear, colorless solution. Plus, phosphoric acid is commonly used in food processing, dental cements, rust removal, and as a catalyst in various chemical reactions. Its mild acidic nature makes it safer to handle compared to strong acids like hydrochloric acid or sulfuric acid.
Magnesium Hydroxide (Mg(OH)2)
Magnesium hydroxide is an inorganic compound with the formula Mg(OH)2. Which means it is commonly known as milk of magnesia when suspended in water. This white solid is only slightly soluble in water but exhibits strong basic properties. Magnesium hydroxide acts as a base capable of accepting protons from acids to form water and salt compounds. It is widely used as an antacid to neutralize stomach acid, as a laxative, and as a flame retardant in various materials. The compound has a molar mass of 58.32 g/mol and decomposes at approximately 350°C Not complicated — just consistent..
The Chemical Equation
The reaction between phosphoric acid and magnesium hydroxide produces magnesium phosphate and water. The unbalanced chemical equation can be written as:
H3PO4 + Mg(OH)2 → Mg3(PO4)2 + H2O
This equation shows the transformation of acidic and basic reactants into salt and water products, characteristic of neutralization reactions. On the flip side, this equation is not balanced, meaning it does not obey the law of conservation of mass, which states that atoms cannot be created or destroyed in a chemical reaction Simple as that..
Balancing the Equation
To balance the chemical equation, we must see to it that the number of each type of atom is equal on both the reactant and product sides. Let us work through this systematically:
Step 1: Count Atoms on Each Side
Reactant side:
- H: 3 (from H3PO4) + 2 (from Mg(OH)2) = 5 hydrogen atoms
- P: 1 phosphorus atom
- O: 4 (from H3PO4) + 2 (from Mg(OH)2) = 6 oxygen atoms
- Mg: 1 magnesium atom
Product side:
- Mg: 3 magnesium atoms
- P: 2 phosphorus atoms
- O: 8 (from Mg3(PO4)2) + 1 (from H2O) = 9 oxygen atoms
- H: 2 hydrogen atoms
Step 2: Balance the Equation
We need to adjust coefficients to equalize all atoms:
-
Start by balancing magnesium (Mg): Add coefficient 3 to Mg(OH)2 H3PO4 + 3 Mg(OH)2 → Mg3(PO4)2 + H2O
-
Balance phosphorus (P): Add coefficient 2 to H3PO4 2 H3PO4 + 3 Mg(OH)2 → Mg3(PO4)2 + H2O
-
Count hydrogen now:
- Reactant side: (2 × 3) + (3 × 2) = 6 + 6 = 12 hydrogen atoms
- Product side: 2 hydrogen atoms (from H2O)
Add coefficient 6 to H2O: 2 H3PO4 + 3 Mg(OH)2 → Mg3(PO4)2 + 6 H2O
-
Verify oxygen balance:
- Reactant side: (2 × 4) + (3 × 2) = 8 + 6 = 14 oxygen atoms
- Product side: (8) + (6 × 1) = 8 + 6 = 14 oxygen atoms ✓
The Balanced Equation
2 H3PO4 + 3 Mg(OH)2 → Mg3(PO4)2 + 6 H2O
This balanced equation now accurately represents the neutralization reaction between phosphoric acid and magnesium hydroxide Surprisingly effective..
Scientific Explanation
This reaction is classified as a double displacement reaction (also known as metathesis) and specifically as an acid-base neutralization. On the flip side, in this process, the hydrogen ions (H+) from phosphoric acid combine with the hydroxide ions (OH-) from magnesium hydroxide to form water molecules. Simultaneously, the phosphate ions (PO4³-) from phosphoric acid combine with the magnesium ions (Mg²+) from magnesium hydroxide to form magnesium phosphate.
The reaction proceeds in steps due to the triprotic nature of phosphoric acid. So each hydrogen atom is removed sequentially as the base neutralizes the acid. That's why magnesium hydroxide, being a strong base despite its limited solubility, effectively removes the acidic protons to form water. The resulting magnesium phosphate is an ionic compound consisting of Mg²+ cations and PO4³- anions arranged in a crystalline lattice structure And it works..
Properties of Products
Magnesium Phosphate (Mg3(PO4)2)
Magnesium phosphate is an inorganic salt that appears as a white crystalline solid. It is relatively insoluble in water but can dissolve in acidic solutions. And this compound contains essential nutrients—magnesium and phosphorus—that are vital for plant and animal metabolism. Still, in the pharmaceutical industry, magnesium phosphate is sometimes used as a dietary supplement. The compound has various polymorphs and can form different hydrate structures depending on preparation conditions.
This is where a lot of people lose the thread.
Water (H2O)
Water is the universal solvent and essential product of all acid-base neutralization reactions. In this reaction, six water molecules are produced for every two phosphoric acid and three magnesium hydroxide molecules that react. The formation of water is the driving force behind the reaction, as it represents a stable, low-energy product Small thing, real impact. Surprisingly effective..
Applications and Uses
The reaction between H3PO4 and Mg(OH)2 has several practical applications:
-
pH Regulation: This reaction is employed in buffer systems to maintain stable pH levels in various industrial processes.
-
Fertilizer Production: Magnesium phosphate can serve as a source of phosphorus and magnesium for agricultural applications The details matter here..
-
Flame Retardant Materials: The reaction products find use in developing fire-resistant materials.
-
Laboratory demonstrations: This reaction illustrates fundamental chemistry concepts for educational purposes.
-
Water Treatment: The reaction helps in removing phosphate ions from wastewater through precipitation.
Safety Considerations
When working with phosphoric acid and magnesium hydroxide, appropriate safety precautions must be observed:
-
Phosphoric acid: Although weaker than strong acids, it can still cause skin irritation and eye damage. Handle with gloves and safety goggles.
-
Magnesium hydroxide: Generally safe in dilute forms (such as milk of magnesia), but concentrated forms can cause irritation.
-
Reaction mixture: The resulting solution may be neutral or slightly basic, depending on stoichiometric ratios The details matter here..
-
Always work in a well-ventilated area and have appropriate spill containment materials available.
Frequently Asked Questions
Why does this reaction produce water?
Water is produced because the H+ ions from the acid combine with OH- ions from the base in a neutralization process. This is a fundamental characteristic of all acid-base reactions.
Is this reaction reversible?
Like most chemical reactions, the equilibrium can be influenced by concentration, temperature, and pressure conditions. On the flip side, the formation of water as a product drives the reaction forward Not complicated — just consistent..
What type of reaction is this?
We're talking about a double displacement reaction and specifically an acid-base neutralization reaction.
Can this reaction occur at room temperature?
Yes, the reaction can proceed at room temperature, though heating may accelerate the reaction rate Took long enough..
What is the practical importance of magnesium phosphate?
Magnesium phosphate serves as a source of essential nutrients in fertilizers and dietary supplements, and it has applications in various industrial processes.
Conclusion
The balanced chemical equation 2 H3PO4 + 3 Mg(OH)2 → Mg3(PO4)2 + 6 H2O represents a classic acid-base neutralization reaction with significant educational and practical value. Practically speaking, understanding this reaction provides insight into fundamental chemical principles including stoichiometry, double displacement reactions, and the behavior of acids and bases. The products—magnesium phosphate and water—have valuable applications across multiple industries, from agriculture to pharmaceuticals. This reaction exemplifies how basic chemistry concepts translate into real-world applications, making it an essential topic for students and professionals alike to understand thoroughly But it adds up..