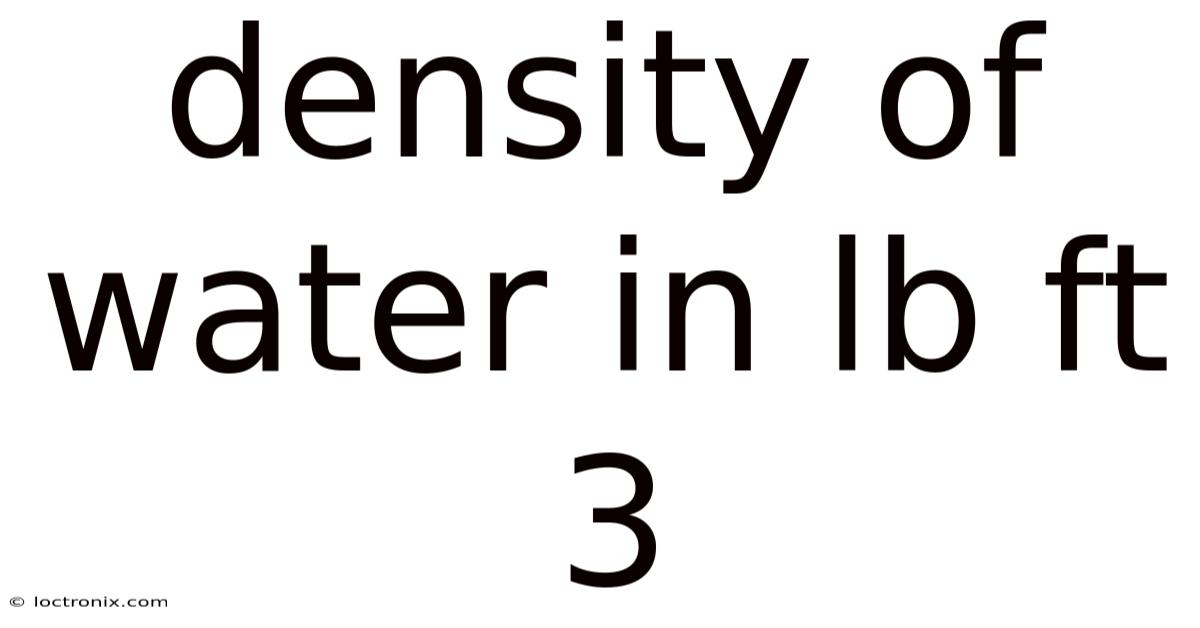Density of Water in lb ft³: A complete walkthrough
The density of water in lb ft³ is a fundamental concept in physics, engineering, and environmental science. For water, this value is typically expressed in pounds per cubic foot (lb ft³), a unit commonly used in the United States and other regions that follow the imperial system. Understanding this measurement is essential for applications ranging from construction and fluid dynamics to climate studies and industrial processes. In real terms, density, defined as mass per unit volume, is a critical property that influences how water interacts with its surroundings. This article explores the significance of water density, how it is calculated, and why it matters in real-world scenarios.
What is Density?
At its core, density quantifies how much mass is contained within a given volume. So for water, this translates to how tightly its molecules are packed. The density of water in lb ft³ is a standardized value that serves as a reference point for comparing other substances.
No fluff here — just what actually works.
Density = Mass ÷ Volume
When applied to water, this formula yields a consistent value under standard conditions. On the flip side, it is important to note that water’s density can fluctuate slightly depending on temperature and pressure. For most practical purposes, the density of water in lb ft³ is accepted as 62.On the flip side, 4 lb ft³ at 4°C (39. 2°F), which is water’s maximum density.
Why Measure Density of Water in lb ft³?
The choice of lb ft³ as a unit for water density stems from its widespread use in industries and engineering fields in countries that adopt the imperial system. This unit is particularly useful in scenarios where large volumes of water are involved, such as in hydrology, civil engineering, or maritime applications. Take this case: knowing the density of water in lb ft³ helps engineers design ships, dams, or pipelines by ensuring they can withstand the forces exerted by water.
Additionally, this measurement is vital for buoyancy calculations. Archimedes’ principle states that an object submerged in a fluid experiences an upward force equal to the weight of the fluid displaced. Since water’s density is a key factor in this principle, engineers and scientists rely on the density of water in lb ft³ to predict whether objects will float or sink.
How to Calculate Density of Water in lb ft³
Calculating the density of water in lb ft³ involves measuring its mass and volume under controlled conditions. Here’s a step-by-step breakdown:
- Measure Mass: Use a scale to determine the weight of a known volume of water. To give you an idea, if you have 1 cubic foot of water, its mass should be approximately 62.4 pounds.
- Measure Volume: Ensure the volume is accurately measured in cubic feet. This can be done using graduated containers or displacement methods.
- Apply the Formula: Divide the mass (in pounds) by the volume (in cubic feet) to obtain the density.
To give you an idea, if 1 cubic foot of water weighs 62.4 pounds, the calculation is straightforward:
Density = 62.4 lb ÷ 1 ft³ = 62.4 lb ft³
This value remains relatively stable under standard atmospheric pressure and temperature. On the flip side, deviations occur when water is heated or cooled, as discussed in the next section.
Scientific Explanation of Water’s Density
The **
Thus, density remains a cornerstone of scientific inquiry, driving progress across disciplines. That's why its precision shapes technologies, informs natural phenomena, and underpins further exploration, ensuring its enduring significance. Such insights continue to illuminate pathways forward, bridging knowledge and application. In closing, it stands as a testament to the interplay between fundamental principles and real-world impact, anchoring understanding in the tangible world.
Scientific Explanation of Water’s Density
The anomalous density behavior of water is fundamentally rooted in its molecular structure and hydrogen bonding. On the flip side, 2°F)**, this structuring reaches an optimal point where molecules are packed most efficiently, yielding maximum density. In liquid water, molecules are in constant motion, allowing them to pack relatively closely. As temperature decreases, molecular kinetic energy reduces, and hydrogen bonds between water molecules become more stable and structured. But below this temperature, the hydrogen bonds begin to form a more open, crystalline lattice—similar to ice—causing the volume to expand and density to decrease. At **4°C (39.This is why ice floats on liquid water, a rare and ecologically critical property that insulates aquatic life during freezing conditions.
Factors such as salinity, pressure, and dissolved impurities also alter density. Seawater, for instance, is denser than freshwater due to dissolved salts, a principle exploited in ocean circulation and marine engineering. Here's the thing — pressure compresses water slightly, increasing its density, though the effect is minimal under most surface conditions. Understanding these nuances allows for accurate modeling in fields from climatology to chemical processing.
Conclusion
The density of water, expressed practically in units like lb ft³, serves as a fundamental bridge between abstract scientific principles and tangible engineering solutions. 4 lb ft³** at peak density—is more than a number; it is a key that unlocks the design of maritime vessels, the management of water resources, and the prediction of natural phenomena. Still, its precision shapes technologies, informs natural phenomena, and underpins further exploration, ensuring its enduring significance. Such insights continue to illuminate pathways forward, bridging knowledge and application. Its value—approximately **62.The underlying science, from hydrogen bonding to thermal expansion, reveals water’s unique role in Earth’s systems, supporting life and shaping climates. But thus, density remains a cornerstone of scientific inquiry, driving progress across disciplines. In closing, it stands as a testament to the interplay between fundamental principles and real-world impact, anchoring understanding in the tangible world.
Conclusion
The density of water, expressed practically in units like lb ft³, serves as a fundamental bridge between abstract scientific principles and tangible engineering solutions. Its value—approximately 62.4 lb ft³ at peak density—is more than a number; it is a key that unlocks the design of maritime vessels, the management of water resources, and the prediction of natural phenomena. The underlying science, from hydrogen bonding to thermal expansion, reveals water’s unique role in Earth’s systems, supporting life and shaping climates. Thus, density remains a cornerstone of scientific inquiry, driving progress across disciplines. Its precision shapes technologies, informs natural phenomena, and underpins further exploration, ensuring its enduring significance. Such insights continue to illuminate pathways forward, bridging knowledge and application. In closing, it stands as a testament to the interplay between fundamental principles and real-world impact, anchoring understanding in the tangible world Took long enough..
The continued study and refinement of our understanding of water’s density are crucial for addressing contemporary challenges. Plus, from developing more efficient desalination technologies to predicting the impact of climate change on water availability, accurate density data and its implications will remain very important. Because of that, further research into the complex interplay of factors influencing water density, particularly in extreme environments, promises to tap into even greater potential for innovation and a deeper appreciation for this vital resource. The story of water's density is far from over; it's an ongoing narrative of scientific discovery and its profound influence on the world around us It's one of those things that adds up..
Continuing smoothly from the established significance of water density:
This foundational understanding propels innovation into emerging frontiers. Engineers are increasingly leveraging precise density variations to design next-generation submersibles capable of navigating extreme ocean depths with unprecedented efficiency, where even minor density changes impact buoyancy and structural stress. Similarly, in environmental science, sophisticated climate models now integrate highly granular data on seawater density shifts to refine predictions of thermohaline circulation patterns, crucial for forecasting regional climate impacts and ocean heat distribution. To build on this, advancements in material science draw inspiration from water's density anomalies, leading to the development of novel hydrogels and smart fluids that mimic water's unique expansion behavior for applications in thermal regulation and drug delivery systems Nothing fancy..
The practical deployment of density knowledge extends to critical infrastructure management. Because of that, modern reservoir operation systems put to use real-time density monitoring, coupled with temperature and salinity sensors, to optimize water release strategies for hydropower generation, irrigation, and flood control, ensuring maximum efficiency while mitigating ecological disruption. In the realm of renewable energy, the principles governing density-driven ocean currents are informing the design of more efficient underwater turbines, harnessing natural kinetic power with minimal environmental footprint. Even in aerospace, the precise calculation of water density is vital for life support system design in spacecraft and habitats, ensuring reliable water storage and recycling in microgravity environments where fluid behavior deviates significantly from Earth's norms.
Conclusion
The density of water, fundamentally anchored at 62.4 lb/ft³, transcends its simple numerical value to become an indispensable axis upon which scientific understanding and technological advancement rotate. Its influence permeates the depths of the oceans, the dynamics of the atmosphere, the core of engineering design, and the detailed machinery of life itself. As we confront escalating global challenges—from climate change adaptation and sustainable resource management to deep-sea exploration and space habitation—the precise comprehension and application of water's density remain key. Plus, it is not merely a static property, but a dynamic variable whose subtle shifts dictate outcomes in natural systems and engineered solutions alike. In practice, the ongoing journey of discovery into water's density is intrinsically linked to our ability to innovate responsibly and deal with the complexities of our planet and beyond. Its enduring significance lies in this profound, tangible connection between a fundamental physical constant and the evolving tapestry of human endeavor and planetary health.