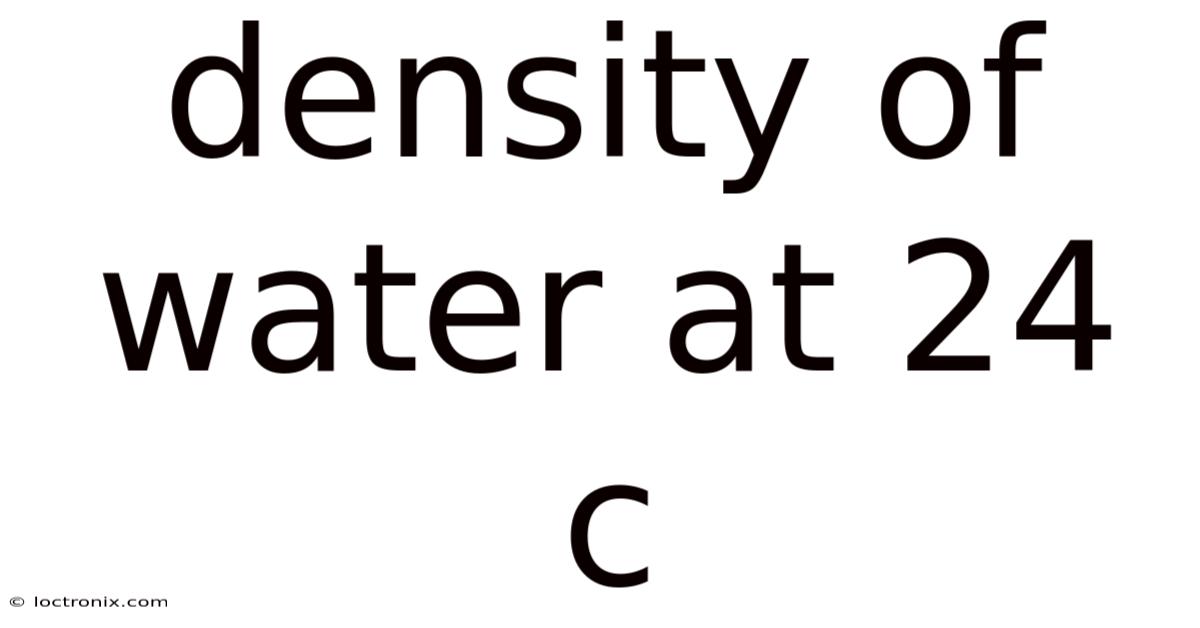The Density of Water at 24°C: Understanding Its Properties and Significance
Water is a unique substance with properties that defy common expectations. One of its most intriguing characteristics is how its density changes with temperature. At 24°C, water exhibits a specific density that plays a critical role in various natural and industrial processes. This article explores the density of water at 24°C, its scientific basis, and its broader implications.
What Is Density?
Density is a measure of mass per unit volume, typically expressed in grams per cubic centimeter (g/cm³) or kilograms per cubic meter (kg/m³). For water, this value is influenced by temperature, pressure, and the presence of dissolved substances. At standard atmospheric pressure, water’s density is a key factor in its behavior in the environment Easy to understand, harder to ignore..
How Does Temperature Affect Water’s Density?
Water’s density does not behave like most substances. Which means while most liquids become less dense as they are heated, water reaches its maximum density at 4°C. Beyond this temperature, its density decreases as the molecules gain energy and move more freely. At 24°C, water is slightly less dense than at 4°C, but still significantly denser than ice, which is less dense due to its crystalline structure And it works..
The Density of Water at 24°C
At 24°C, the density of pure water is approximately 0.Now, 997 g/cm³ or 997 kg/m³. This value is slightly lower than the density at 4°C (1.On the flip side, 000 g/cm³) but still much higher than the density of ice (0. That's why 917 g/cm³). Plus, the decrease in density with increasing temperature is due to the weakening of hydrogen bonds between water molecules. As temperature rises, these bonds break more frequently, allowing molecules to spread out and occupy more space.
No fluff here — just what actually works.
Why Does Water’s Density Peak at 4°C?
Water’s unusual density behavior is tied to its molecular structure. In practice, hydrogen bonds create a lattice-like arrangement in liquid water, which becomes more ordered as temperature decreases. Practically speaking, at 4°C, this arrangement is most efficient, maximizing density. On the flip side, as temperature continues to drop below 4°C, the molecules begin to form ice crystals, which are less dense because the hydrogen bonds are arranged in a more open, hexagonal structure. This is why ice floats on water.
Comparing Densities at Different Temperatures
To better understand the significance of 24°C, consider the following comparisons:
- At 0°C (freezing point): 0.9998 g/cm³
- At 4°C: 1.000 g/cm³ (maximum density)
- At 20°C: 0.998 g/cm³
- At 24°C: 0.
At 100°C (boiling point), water’s density further decreases to approximately 0.958 g/cm³, reflecting its transition into steam as it absorbs heat. Practically speaking, this stark contrast between the density at 24°C and the boiling point underscores how temperature profoundly influences water’s physical state and properties. The gradual decline in density from 4°C to 100°C illustrates a consistent trend, where increased thermal energy disrupts hydrogen bonding, allowing molecules to spread apart Worth knowing..
Practical Implications of Water’s Density at 24°C
The density of water at 24°C has significant real-world applications. In aquatic ecosystems, this density plays a role in thermocline formation—the layer in a body of water where temperature and density change rapidly with depth. At 24°C, water is less dense than colder layers, contributing to stratification in lakes and oceans, which affects nutrient distribution and marine life. In industrial settings, precise knowledge of water’s density at specific temperatures is critical for processes like cooling systems, where maintaining optimal density ensures efficient heat transfer. Additionally, in hydrology, understanding density variations helps model groundwater flow and predict flood risks, as denser water sinks and influences aquifer dynamics No workaround needed..
The Broader Significance
Water’s unique density behavior, particularly its maximum at 4°C and the subsequent decrease at higher temperatures, is a cornerstone of Earth’s climate and ecological systems. This anomaly, rooted in hydrogen bonding, ensures that aquatic environments remain habitable even in freezing conditions, as ice forms on the surface while liquid water persists below. The density at 24°C, while not the peak, still reflects water’s adaptability and resilience. It highlights how a single substance can exhibit behavior that defies intuition, shaping everything from weather patterns to industrial engineering.
All in all, the density of water at 24°C is more than a numerical value; it is a testament to the detailed balance of molecular interactions that define this essential substance. By studying such properties, scientists gain insights into natural processes and develop technologies that harness water’s unique characteristics. As climate change and environmental challenges grow, understanding these fundamental properties becomes increasingly vital, reminding us that even the most common elements can hold extraordinary secrets.
You'll probably want to bookmark this section.
The interplay of matter and environment continues to evolve, demanding constant attention. Such considerations anchor our understanding firmly within the natural order.
In synthesizing these insights, clarity emerges as a guiding force. The journey underscores a universal truth: mastery of the fundamentals reveals broader implications.
Thus, concluding with reflection, the study persists as a vital thread in the tapestry of knowledge, inviting further exploration and mindful application.
Conclusion: Water’s properties, though nuanced, remain indispensable, shaping our world both implicitly and explicitly. Their study fosters awareness that permeates every facet of existence, reminding us to cherish and apply this knowledge thoughtfully But it adds up..