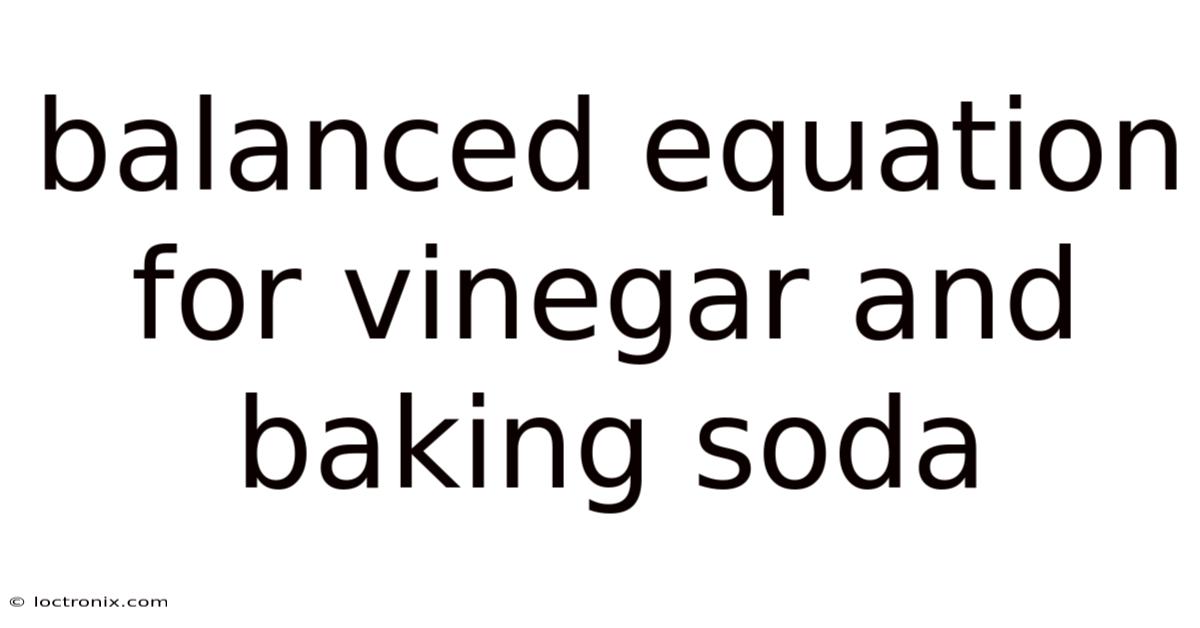The Science Behind the Classic Reaction: Vinegar and Baking Soda
When it comes to household experiments that are as simple as they are spectacular, the combination of vinegar and baking soda stands out. This seemingly innocent pairing can create a delightful chemical reaction that not only entertains but also educates. In this article, we'll dive into the science behind the balanced equation for vinegar and baking soda, exploring what happens when these two common substances come into contact, and why it's such a popular choice for science demonstrations and DIY projects.
Introduction to the Reaction
The reaction between vinegar and baking soda is a classic example of an acid-base reaction. Here's the thing — vinegar, which is primarily composed of acetic acid (CH₃COOH), reacts with baking soda, which is sodium bicarbonate (NaHCO₃). When these two substances mix, they produce carbon dioxide gas (CO₂), water (H₂O), and sodium acetate (NaCH₃COO). This reaction is not only visually appealing, with the fizzing and bubbling that accompanies the release of carbon dioxide, but it also has practical applications in cooking, cleaning, and even as a simple science experiment.
The Balanced Chemical Equation
The balanced chemical equation for the reaction between vinegar and baking soda is as follows:
[ \text{CH₃COOH (acetic acid)} + \text{NaHCO₃ (sodium bicarbonate)} \rightarrow \text{CO₂ (carbon dioxide)} + \text{H₂O (water)} + \text{NaCH₃COO (sodium acetate)} ]
This equation shows that one molecule of acetic acid reacts with one molecule of sodium bicarbonate to produce one molecule of carbon dioxide, one molecule of water, and one molecule of sodium acetate. The reaction is balanced because the number of atoms of each element on the reactant side of the equation is equal to the number of atoms of that element on the product side.
The Science Explained
Acid-Base Reaction
The reaction between vinegar and baking soda is an acid-base reaction. Acids are substances that can donate protons (H⁺ ions), and bases are substances that can accept protons. Consider this: in this case, the acetic acid in vinegar donates a proton to the bicarbonate ion in baking soda, which then becomes a carbonate ion. This process is known as neutralization Not complicated — just consistent. Took long enough..
Production of Carbon Dioxide
The carbonate ion then reacts with water to produce hydrogen carbonate ions, which further dissociate into bicarbonate ions and hydrogen ions. The hydrogen ions then react with the bicarbonate ions to form carbonic acid (H₂CO₃), which is unstable and quickly decomposes into carbon dioxide gas and water. This is the source of the bubbling and fizzing you see when vinegar and baking soda mix.
The Role of Temperature
The rate at which this reaction occurs can be influenced by temperature. Warmer temperatures can increase the rate of reaction, as the molecules have more energy to collide and react. This is why the reaction might be faster when vinegar and baking soda are mixed in warm water.
Practical Applications
The reaction between vinegar and baking soda has several practical applications:
- Cleaning: The fizzing action can help to scrub away grime and break down organic matter.
- Cooking: In baking, the reaction between vinegar and baking soda can help to leaven the dough, creating a lighter texture.
- DIY Experiments: The reaction is a popular choice for science experiments because it is safe, easy to observe, and can be scaled up for dramatic effect.
Frequently Asked Questions
Can I use other acids instead of vinegar?
Yes, you can use other acids such as citric acid or hydrochloric acid to react with baking soda. That said, vinegar is preferred for its mildness and the fact that it produces a lot of carbon dioxide, which is the most visually striking part of the reaction Worth keeping that in mind..
Is this reaction safe?
Yes, the reaction between vinegar and baking soda is safe for most people. That said, it's always important to wear gloves and goggles when handling chemicals, even if they are common household items Easy to understand, harder to ignore..
Can I use vinegar and baking soda to clean my oven?
Yes, vinegar and baking soda can be used to clean ovens. The reaction can help to break down grease and grime, and the baking soda can act as a mild abrasive It's one of those things that adds up..
Conclusion
The reaction between vinegar and baking soda is a perfect example of how simple ingredients can produce a complex and fascinating chemical reaction. By understanding the balanced equation and the science behind the reaction, you can appreciate the beauty of chemistry in your everyday life. Whether you're a student learning about acid-base reactions or a DIY enthusiast looking for a fun project, the vinegar and baking soda reaction is a great way to explore the wonders of chemistry.
The interplay between elements reveals a dance of chemistry underpinning daily life.
Final Reflection
Understanding such processes deepens our grasp of nature’s layered systems, bridging science with practicality. Such discoveries remind us of the profound connections linking us to the world around us Worth keeping that in mind..
In closing, the simplicity of reactions often masks their significance, urging us to seek clarity and curiosity in every interaction.
Temperature not only accelerates the reaction but also shifts the balance of dissolved gases, subtly shaping the foam’s stability and the distribution of residual sodium acetate. This sensitivity to heat finds use in portable warmers and improvised cold packs, where the same mixture can either release or absorb energy depending on how ingredients are combined and insulated Easy to understand, harder to ignore. But it adds up..
Beyond cleaning and baking, the controlled release of carbon dioxide supports fire suppression in small-scale demonstrations, inflates air pockets in lightweight doughs without metallic aftertaste, and powers miniature rockets that illustrate thrust and conservation of momentum. Even in wastewater treatment, analogous acid–carbonate reactions help modulate pH and precipitate metals, showing how a kitchen staple can echo industrial principles Surprisingly effective..
As these examples accumulate, they reinforce a broader truth: everyday reactions serve as gateways to systems thinking. By observing how modest changes in concentration, temperature, or surface contact ripple outward, we cultivate habits of mind that translate to larger challenges—resource efficiency, safety, and innovation.
In the end, vinegar and baking soda do more than bubble; they invite us to witness transformation with care and imagination. Through such humble experiments, curiosity matures into insight, reminding us that the most enduring advances often begin with a simple, honest question and the willingness to see it through.
People argue about this. Here's where I land on it.
...the resulting sodium acetate solution can be carefully isolated and repurposed. Its ability to absorb and release thermal energy makes it an excellent medium for regulating temperature in reusable heat pads, demonstrating how a common household reaction can be engineered for practical, sustainable applications.
This versatility extends into educational realms, where the reaction serves as a tangible bridge between abstract theory and hands-on learning. Students can measure gas production, calculate reaction rates, and visualize stoichiometry, transforming a simple kitchen experiment into a profound lesson on conservation of mass and energy. The tangible evidence of a gas bubble or a temperature shift makes the invisible world of molecules feel immediate and real Took long enough..
The bottom line: the enduring appeal of this reaction lies in its accessibility and depth. On the flip side, it requires nothing more than a pantry staple and a moment of curiosity, yet it opens a window into fundamental principles of chemistry, physics, and engineering. By embracing this simple interaction, we not only solve immediate practical problems but also cultivate a mindset of inquiry and resourcefulness. In doing so, we honor the quiet power of basic science to illuminate and inspire.