Atomic Mass Of An Element Is Equal To
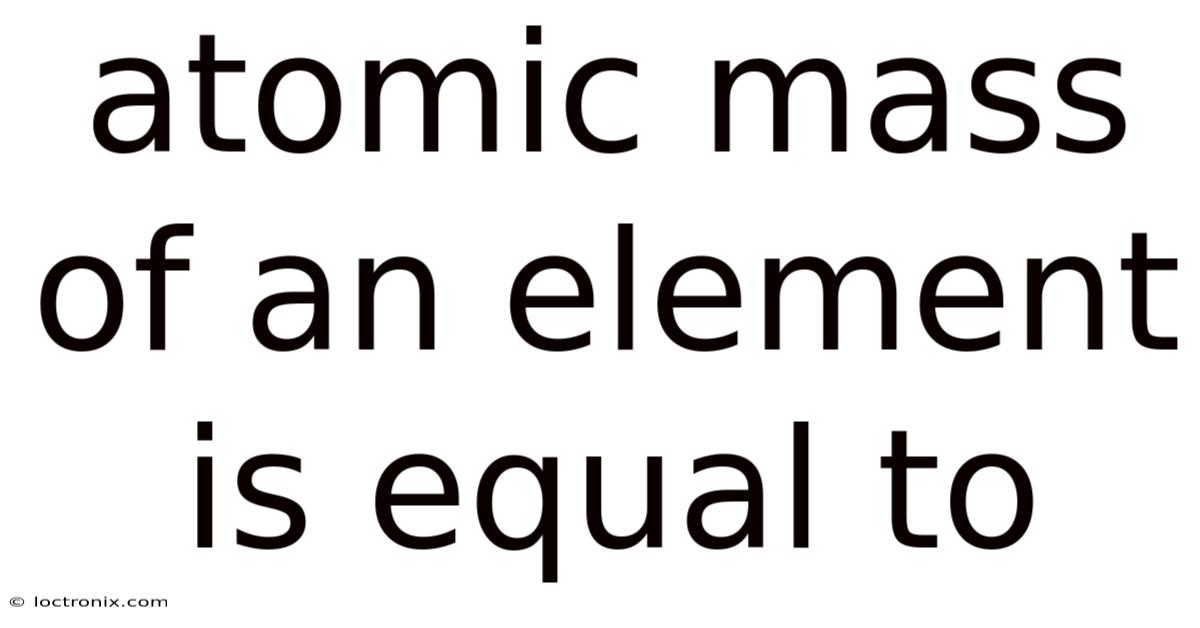
The atomic mass ofan element is equal to the weighted average of the masses of all its naturally occurring isotopes, expressed in atomic mass units (u).
This simple statement hides a wealth of nuance that underpins everything from chemical bonding to nuclear physics. Understanding what the atomic mass truly represents, how it is calculated, and why it matters can transform a vague classroom fact into a powerful mental model. In this article we will explore the concept step by step, clarify common misconceptions, and highlight practical applications—all while keeping the explanation accessible to students, educators, and curious readers alike.
What Is Atomic Mass?
At its core, atomic mass (also called atomic weight) quantifies the mass of a single atom of an element on a relative scale. Unlike molar mass, which is expressed in grams per mole, atomic mass is a dimensionless number that compares the mass of an atom to a reference—historically oxygen‑16 and, more recently, carbon‑12.
Key points to remember
- Atomic mass is measured in atomic mass units (u), where 1 u ≈ 1.660539 × 10⁻²⁴ g.
- It reflects the average mass of atoms in a naturally occurring sample, accounting for isotopic abundance.
- The term relative atomic mass is often used interchangeably, emphasizing the comparison to the reference standard.
How Is Atomic Mass Determined?
Calculating the atomic mass involves several systematic steps that blend experimental data with mathematical weighting. Below is a concise roadmap that illustrates the process.
-
Identify all isotopes of the element that exist in nature.
Example: Carbon has two stable isotopes—¹²C and ¹³C. -
Measure the atomic mass of each isotope using techniques such as mass spectrometry.
Result: ¹²C = 12.000 u (by definition), ¹³C ≈ 13.003 u. -
Determine the natural abundance of each isotope, usually expressed as a percentage or fraction.
Typical values: ¹²C ≈ 98.93 %, ¹³C ≈ 1.07 %. -
Convert abundances to fractional form (divide by 100). 5. Multiply each isotope’s mass by its fractional abundance.
-
Sum the products to obtain the weighted average, which is the atomic mass of the element. #### Example Calculation for Carbon
| Isotope | Mass (u) | Fractional Abundance | Product |
|---|---|---|---|
| ¹²C | 12.000 | 0.9893 | 11.8716 |
| ¹³C | 13.003 | 0.0107 | 0.1391 |
| Total | — | — | 12.0107 u |
The final value, ≈ 12.01 u, is the atomic mass of carbon that appears on the periodic table.
Factors That Influence Atomic Mass
Several variables can affect the reported atomic mass of an element, especially when dealing with synthetic or short‑lived isotopes.
- Isotopic composition: Elements with multiple stable isotopes (e.g., chlorine, bromine) show noticeable variations in atomic mass depending on geographic source.
- Sample purity: Artificially enriched samples can shift the weighted average, leading to slightly different atomic masses.
- Measurement precision: Advances in instrumentation improve accuracy, occasionally refining published atomic masses.
Typical sources of variation
- Natural geological samples may contain trace amounts of rare isotopes.
- Laboratory‑produced isotopes often have different abundances than those found in nature.
Common Misconceptions and FAQs
FAQ 1: Is atomic mass the same as atomic number?
No. The atomic number counts only the protons in the nucleus, while atomic mass includes both protons and neutrons (and, to a negligible extent, the mass of electrons).
FAQ 2: Why does the periodic table show a non‑integer atomic mass?
Because the listed value is a weighted average of all naturally occurring isotopes. Only a pure isotope (e.g., ¹²C) has an integer mass; mixtures yield fractional results.
FAQ 3: Can atomic mass change over time?
In principle, yes. If the isotopic composition of a sample shifts—through radioactive decay, enrichment, or contamination—the atomic mass will adjust accordingly. However, for most stable elements the change is imperceptibly small on human timescales.
FAQ 4: How does atomic mass relate to molar mass?
Molar mass (grams per mole) is numerically equal to atomic mass when expressed in atomic mass units. For example, carbon’s atomic mass of 12.01 u corresponds to a molar mass of approximately 12.01 g mol⁻¹.
FAQ 5: What role does relative atomic mass play in chemistry?
It allows chemists to perform stoichiometric calculations without dealing with absolute masses. By using the relative scale, reaction equations can be balanced using whole numbers that reflect the proportion of atoms involved.
Why Understanding Atomic Mass Matters
Grasping the concept of atomic mass does more than satisfy academic curiosity; it equips learners with a foundational tool for interpreting the physical world.
- Chemical reactions: Knowing atomic masses enables balanced equations, limiting‑reactant calculations, and yield predictions.
- Spectroscopy: Isotopic masses affect vibrational frequencies, influencing
infrared and Raman spectra, which are essential for identifying molecular structures.
- Nuclear science: Precise atomic masses are crucial for calculating binding energies, reaction Q-values, and decay pathways.
- Materials science: Isotopic composition can influence material properties, such as thermal conductivity in semiconductors or the behavior of superconductors.
In essence, atomic mass is not merely a number on the periodic table—it is a gateway to understanding the quantitative relationships that govern matter at its most fundamental level. By appreciating its nuances, from isotopic variations to its role in chemical calculations, students and professionals alike can approach scientific problems with greater precision and insight. Whether predicting the outcome of a reaction, analyzing a spectroscopic dataset, or designing a new material, the concept of atomic mass remains an indispensable cornerstone of modern chemistry and physics.
...and even the development of quantum mechanical models that describe nuclear structure. As measurement techniques grow more precise—with instruments like mass spectrometers capable of distinguishing minute mass differences between isotopes—our understanding of atomic mass continues to deepen, revealing subtle effects such as nuclear binding energy variations and the influence of electron configuration on mass.
Ultimately, the atomic mass listed on the periodic table is far more than a static figure; it is a dynamic synthesis of natural abundance, nuclear stability, and fundamental physical laws. It connects the discrete world of individual atoms to the continuous realm of measurable matter, serving as a critical translator between the microscopic and macroscopic scales. For anyone engaged in the quantitative sciences, mastering this concept is not optional—it is essential. It empowers accurate prediction, rigorous analysis, and innovative design across disciplines. As we probe deeper into the fabric of the universe, from the heart of stars to the intricacies of synthetic molecules, the humble value of atomic mass remains a quiet but powerful constant, reminding us that even the most abstract numbers carry the weight of reality.
The enduring relevance of atomic mass lies in its ability to bridge the gap between abstract theory and tangible application. As we continue to refine our measurement techniques, from quantum computing simulations to next-generation mass spectrometry, the precision with which we can quantify atomic mass will undoubtedly unlock new frontiers in science. For instance, in medicine, isotopic labeling using precise atomic mass data enables advanced diagnostic tools and targeted therapies. In environmental science, understanding isotopic shifts in atmospheric or geological samples aids in tracking climate change and ancient Earth processes. Even in the realm of artificial intelligence, atomic mass data could inform machine learning models designed to predict material behavior or chemical reactivity with unprecedented accuracy.
This concept also serves as a reminder of the interconnectedness of scientific disciplines. The same atomic mass values that underpin a chemist’s balanced equation might inform a physicist’s study of stellar nucleosynthesis or an engineer’s design of a nuclear reactor. It is a testament to the universality of fundamental principles—how a single, seemingly simple parameter can resonate across scales and specializations.
In a world increasingly driven by data and precision, mastering atomic mass is not just an academic exercise; it is a practical necessity. It equips us to navigate the complexities of modern challenges, whether in developing sustainable energy sources, combating diseases, or exploring the cosmos. As our curiosity about the universe’s deepest mysteries grows, so too will our reliance on the quiet yet profound insights that atomic mass provides. Ultimately, it is more than a number—it is a lens through which we perceive and shape the material world, a constant that anchors our quest for knowledge across time and disciplines. In embracing its nuances, we honor the intricate dance of matter and energy that defines existence itself.
Latest Posts
Latest Posts
-
6 Characteristics Of All Living Things
Mar 25, 2026
-
Chart Of Fractions Decimals And Percents
Mar 25, 2026
-
Pleiotropy Is Evident When One Gene
Mar 25, 2026
-
What Grades Do You Need To Get Into Clemson
Mar 25, 2026
-
Draw The Starting Structure That Would Lead
Mar 25, 2026