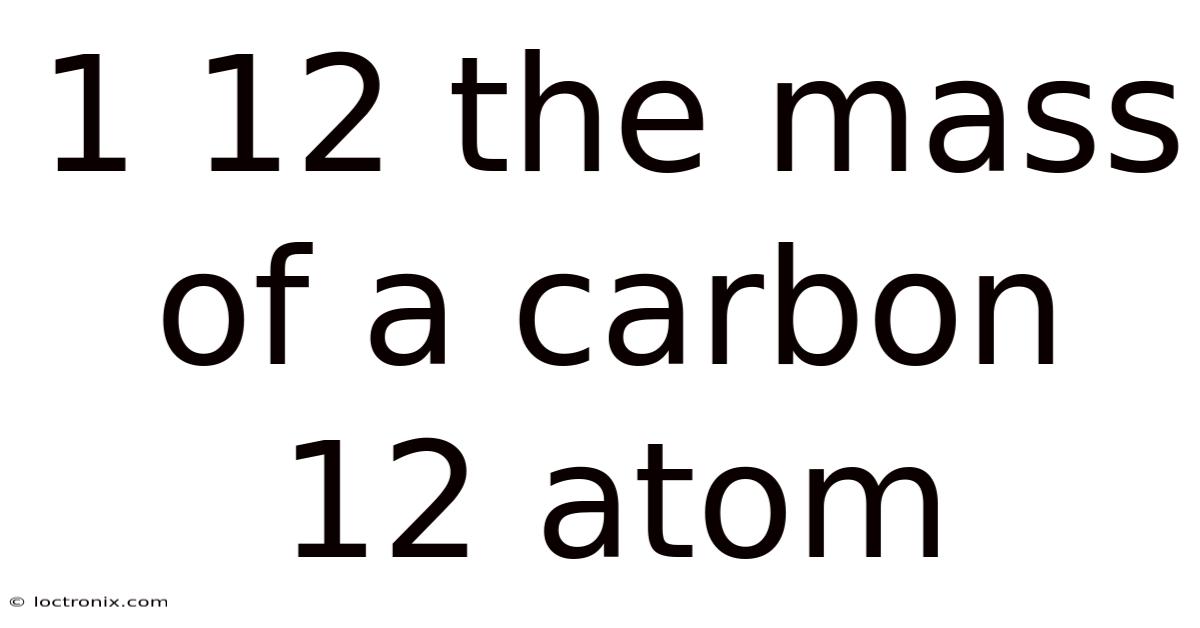The Carbon-12 Atom: Defining the Standard for Atomic Mass
The mass of a single carbon-12 atom stands as one of the most fundamental constants in science, underpinning our understanding of chemistry, physics, and the very building blocks of matter. Day to day, this seemingly simple atom, composed of just six protons, six neutrons, and six electrons, serves as the cornerstone for defining the atomic mass unit (amu), the standard by which all other atomic masses are measured. Because of that, its precise mass isn't just a number; it's the bedrock upon which the periodic table, chemical calculations, and our comprehension of atomic structure rest. Understanding the mass of the carbon-12 atom reveals the complex dance of particles that governs the physical world.
Introduction: The Atomic Mass Unit and Carbon-12's key Role
Imagine trying to weigh individual atoms. And without this precise definition, atomic weights would be mere estimates, and the quantitative predictions of chemistry would be hopelessly imprecise. Defined as exactly 12 atomic mass units (amu), the carbon-12 atom provides the standard against which the masses of all other atoms are compared. Their masses are incomprehensibly small, far beyond the reach of everyday scales. In practice, that reference is the carbon-12 atom. This fundamental challenge in measuring atomic-scale mass was resolved in the mid-20th century by establishing a universal reference point. And this definition, adopted internationally, means that one amu is precisely one-twelfth the mass of a carbon-12 atom. The carbon-12 atom's mass is not arbitrary; it is the carefully calibrated anchor point for the entire field of atomic and molecular science That's the whole idea..
Atomic Structure: The Building Blocks of Carbon-12
To grasp the mass of a carbon-12 atom, we must first understand its composition. That's why at its core lies the nucleus, a compact, dense region containing protons and neutrons. Orbiting this nucleus, at vast distances compared to the nucleus's size, are electrons.
- Protons: Positively charged particles residing in the nucleus. Carbon-12 has 6 protons. The number of protons defines the element itself – carbon.
- Neutrons: Neutral particles also found in the nucleus. Carbon-12 has 6 neutrons.
- Electrons: Negatively charged particles orbiting the nucleus. Carbon-12 has 6 electrons.
The mass of an atom is primarily concentrated in its nucleus, where protons and neutrons reside. That said, electrons contribute negligibly to the total atomic mass due to their minuscule mass. Because of that, the mass of a proton and a neutron is approximately equal, around 1. Still, 67 × 10⁻²⁷ kilograms (or 1 amu), while an electron is about 9. Which means 11 × 10⁻³¹ kilograms, roughly 1/1836th the mass of a proton or neutron. So, for calculating the mass of a carbon-12 atom, we focus almost entirely on the combined mass of its 6 protons and 6 neutrons Still holds up..
Mass Determination: Calculating the Carbon-12 Mass
The mass of a single carbon-12 atom is a fixed, defined value. That said, to understand how it's conceptualized and calculated, we consider the masses of its constituent particles:
- Mass of a Proton: Approximately 1.6726 × 10⁻²⁷ kg or 1.007825 amu.
- Mass of a Neutron: Approximately 1.6749 × 10⁻²⁷ kg or 1.008665 amu.
- Mass of an Electron: Approximately 9.109 × 10⁻³¹ kg or 0.00054858 amu.
For a carbon-12 atom:
- Mass from 6 Protons = 6 × 1.007825 amu = 6.04695 amu
- Mass from 6 Neutrons = 6 × 1.008665 amu = 6.05199 amu
- Mass from 6 Electrons = 6 × 0.00054858 amu ≈ 0.00329148 amu
Total Calculated Mass (Before Binding Energy): 6.04695 amu + 6.05199 amu + 0.00329148 amu ≈ 12.10223148 amu
This calculated mass (approximately 12.10223148 amu) is significantly higher than the actual mass of the carbon-12 atom. This discrepancy arises due to mass defect Most people skip this — try not to. Less friction, more output..
Mass Defect and Binding Energy: The Nuclear Glue
The mass defect is the difference between the calculated mass of the individual nucleons (protons and neutrons) and the actual measured mass of the nucleus they form. The energy released when nucleons bind together to form a nucleus is called the binding energy. This binding energy represents the energy equivalent of the mass defect. On the flip side, this difference is explained by Einstein's famous equation, E=mc². In plain terms, some of the mass of the individual nucleons is converted into the binding energy that holds the nucleus together Worth keeping that in mind..
For carbon-12, the actual measured mass is exactly 12 amu. The calculated mass (12.10223148 amu) minus the actual mass (12.00000 amu) gives a mass defect of approximately 0.10223148 amu. This mass defect is the binding energy of the carbon-12 nucleus, converted into energy.
That's why, the defined mass of the carbon-12 atom is exactly 12 amu. The calculated mass (12.This definition is precise and absolute, not calculated from individual particle masses. So 10223148 amu) is what you would get if you simply added up the masses of 6 protons and 6 neutrons without accounting for the binding energy released during nucleus formation. The actual, measured mass of the bound nucleus is less due to the conversion of mass into binding energy That's the whole idea..
Real talk — this step gets skipped all the time.
Significance: Why Carbon-12 Matters
The choice of carbon-12 as the standard atomic mass unit reference is deeply significant:
- Universal Standard: It provides a single, consistent point of reference for measuring the mass of all atoms and molecules. This allows chemists and physicists worldwide to communicate atomic masses unambiguously.
- Foundation for the Periodic Table: The atomic masses listed on the periodic table are calculated relative to the mass of carbon-12. These masses are crucial for determining
the properties and behavior of elements. That's why Historical Context: The selection of carbon-12 wasn't arbitrary. Here's the thing — 4. Even so, it was chosen because it's a naturally abundant isotope, relatively stable, and its mass is easily reproducible and measurable. 3. But Calibration of Measurement Tools: Carbon-12 is used to calibrate mass spectrometers and other analytical instruments, ensuring accurate measurements across various scientific disciplines. Its well-defined mass provides a reliable benchmark for instrument performance. Without this standard, comparing and understanding chemical reactions and material properties would be immensely more complex. This practical consideration has contributed to its enduring role as the standard Not complicated — just consistent. That alone is useful..
The understanding of mass defect and binding energy revolutionized our understanding of nuclear physics and the fundamental forces governing the universe. It showcased that mass and energy are interchangeable, a cornerstone of modern physics. Carbon-12, as the anchor for atomic mass units, serves as a constant reminder of this profound relationship and the nuanced balance that holds matter together That alone is useful..
So, to summarize, the definition of atomic mass is not simply an arbitrary assignment. Carbon-12, with its precisely defined mass, provides a crucial foundation for chemistry, physics, and countless other scientific endeavors. It’s a carefully constructed system built upon fundamental physics and practical considerations. In real terms, its significance extends far beyond the laboratory, underpinning our understanding of the composition and behavior of matter in the universe. The concept of mass defect and binding energy, demonstrated through the example of carbon-12, highlights the dynamic nature of matter and the profound connection between mass and energy.
This foundational relationship between mass and energy, anchored by carbon-12, extends well beyond theoretical frameworks into the practical realities of modern science and industry. In pharmaceutical research, precise molecular mass calculations dictate drug stability, bioavailability, and metabolic pathways, where even fractional deviations can alter therapeutic outcomes. In real terms, materials engineers rely on exact isotopic mass data to design semiconductors, superconductors, and radiation-resistant alloys, pushing the boundaries of what modern technology can achieve. Meanwhile, astrophysicists use carbon-12 as a cosmic yardstick to model stellar nucleosynthesis, tracing how heavier elements are forged in supernovae and neutron star mergers across billions of years.
As experimental capabilities advance, the atomic mass unit continues to serve as a testing ground for fundamental physics. Practically speaking, modern Penning trap spectrometers and laser-based measurement techniques now determine atomic masses with uncertainties below one part in a billion. Worth adding: these extraordinary levels of precision have uncovered subtle anomalies in nuclear mass predictions, prompting refinements to shell models and offering stringent constraints on theories beyond the Standard Model. On the flip side, in metrology, the carbon-12 standard remains integral to the International System of Units, ensuring that measurements remain consistent across laboratories, industries, and generations. Rather than a static relic, it functions as a living benchmark that evolves alongside our capacity to observe and quantify the natural world.
The bottom line: the atomic mass unit defined by carbon-12 represents more than a numerical convention; it is a testament to the human drive to impose order on complexity. As research ventures into quantum materials, precision medicine, and deep-space exploration, this foundational reference will continue to anchor discovery. Think about it: by grounding abstract nuclear phenomena in a reproducible, universally accepted standard, science has forged a common language that bridges disciplines, eras, and continents. It reminds us that even the most minute measurements can illuminate the grandest structures of reality, proving that clarity, consistency, and shared standards remain the bedrock of scientific progress.